| Issue |
A&A
Volume 549, January 2013
|
|
|---|---|---|
| Article Number | A128 | |
| Number of page(s) | 4 | |
| Section | Atomic, molecular, and nuclear data | |
| DOI | https://doi.org/10.1051/0004-6361/201220375 | |
| Published online | 10 January 2013 | |
The extended spectroscopic database for deuterated species of formamide up to 1 THz ⋆
1
Laboratoire de Physique des Lasers, Atomes, et Molécules, UMR CNRS 8523,
Université de Lille 1,
59655,
Villeneuve d’Ascq Cedex,
France
e-mail: This email address is being protected from spambots. You need JavaScript enabled to view it.
2
Sciences Chimiques de Rennes-École Nationale Supérieure de Chimie
de Rennes-CNRS, 35700
Rennes,
France
Received: 12 September 2012
Accepted: 1 November 2012
Abstract
Context. Formamide (HCONH2) is the simplest molecule with a peptide bond detected in the interstellar medium. One of the keys for understanding the mechanisms for forming formamide in massive hot cores is the comparison of the relative abundances of its deuterated isotopologues.
Aims. No deuterated species of formamide have been detected to date mostly owing to the lack of laboratory spectral data, which is limited by cm-wave measurements up to 40 GHz. Therefore, we decided to extend the measurements and the analysis of the rotational spectra of deuterated istopologues of formamide into the terahertz range.
Methods. We measured the rotational spectra of deuterated formamide in the frequency range 150–950 GHz using the Lille fast-scan and solid-state source spectrometers.
Results. The ground vibrational states of 4 different isotopologues of formamide, DCONH2, cis-HCONHD, trans-HCONHD, and HCOND2, were assigned and analysed. The sets of the rotational and centrifugal distortion constants we obtained allow us to calculate accurate predictions of the transition frequencies for deuterated formamide in the frequency range above 1 THz and at least for J < 65 and Ka < 25.
Key words: ISM: molecules / molecular data / submillimeter: ISM / line: identification
Full Table 2 is only available at the CDS via anonymous ftp to cdsarc.u-strasbg.fr(130.79.128.5) or via http://cdsarc.u-strasbg.fr/viz-bin/qcat?J/A+A/549/A128
Research fellow. Permanent address: V.N. Karazin Kharkov National University, Svobody Sq. 4, 61022 Kharkov, Ukraine.
© ESO, 2013
1. Introduction
Formamide (HCONH2) and acetamide (CH3CONH2) are the only molecules with a peptide bond – C(O)NH – that have been detected in the interstellar medium (Rubin et al. 1971; Hollis et al. 2006; Motiyenko et al. 2012). The peptide bond provides the link for the polymerization of amino acids. The mechanism of synthesis of formamide in the massive hot cores Orion KL and Sgr B2(N) (Turner 1991; Hollis et al. 2006) is not clearly understood. The most probable scenario is that formamide is formed on the grain surface and that its gas-phase abundance reflects recent evaporation of ices due to heating by the newly formed star. Several different reaction pathways, as well as experimental studies of the production of formamide starting from interstellar ice analogs, have recently been performed by Jones et al. (2011). It has been also shown that formamide can be synthesized from the vacuum ultraviolet irradiation of solid HNCO (Raunier et al. 2004) and by the photolysis of solid NH2CH2OH (Bossa et al. 2009). In the gas phase, formamide could form from the reaction between NH2 and H2CO (Garrod et al. 2008), but it seems that such a reaction has a lower rate (Woodall et al. 2007). Ion-molecule processes leading to formamide formation are discussed in the paper by Halfen et al. (2011), in particular, the reaction of formaldehyde and protonated ammonia, creating protonated formamide, followed by dissociative electron recombination.
The relative abundances of the deuterated isotopologues of molecular species can provide interesting information on the mechanisms of its formation. For example, it is believed that methanol is formed on grain surfaces and released in the gas-phase via mantle evaporation. For methanol the abundance ratio of the deuterated isotopologues, [CH2DOH]/[CH3OD], is predicted to be about three (Charnley et al. 1997). However, this ratio has been observed to lie in the range 5 to 30 towards low-mass and intermediate protostars (Parise et al. 2006; Ratajczak et al. 2011) and close to or below one in high-mass protostars (Ratajczak et al. 2011). Recent laboratory studies (Nagaoka et al. 2005; Ratajczak et al. 2009) have shown that a much lower abundance of CH3OD than for CH2DOH, as observed in low- and intermediate-mass protostars (Ratajczak et al. 2011), can be qualitatively explained via H/D exchanges occurring on the grain surface following methanol formation. The results of Ratajczak et al. (2009), in particular, suggest that such exchanges occur on the hydroxyl (–OD) or amine (–ND) functional groups, via hydrogen bonds with water ice. As a result, in the case of formamide, DCONH2 species should be significantly more abundant than HCONDH and HCONHD ones, if solid H/D exchanges do happen, because these should take place on the amino group –ND2. On the other hand, the three deuterated isotopologues of formamide could have a similar abundance owing to some alternative or additional mechanisms in the post-evaporative gas phase, as discussed for methanol by Ratajczak et al. (2011).
Determined rotational and centrifugal distortion parameters.
Formamide is a small molecule with relatively light molecular weight (M = 44) and with non-zero μa = 3.62 D and μb = 0.85 D dipole moment components (Kurland & Wilson 1957). Therefore its rotational spectrum extends in the terahertz range even under the low-temperature conditions of the interstellar medium. The rotational spectrum of the parent species of formamide has been extensively studied up to 1 THz (Kryvda et al. 2009; Motiyenko et al. 2012). The available data on its deuterated isotopologue are much more limited. Only a few lines of different NH2 deuterated species have been observed by Kurland & Wilson (1957) in the frequency range 18–40 GHz. The microwave spectra of ten isotopic species of formamide have been investigated by Costain & Dowling (1960). They measured no more than ten lines for each deuterated species in the frequency range 9–43 GHz. The rotational analysis was limited by determining three rotational constants without taking centrifugal distortion correction into account. Obviously the previously obtained data are sufficient to provide reliable frequency predictions in the range up to 40–50 GHz, but further extrapolation is not possible. In the present paper we report the new extensive analysis of the rotational spectra of all singly substituted D-isotopologues of formamide, as well as of its ND2 species in the frequency range up to 950 GHz.
2. Experimental details
2.1. Synthesis of deuterated species
Trideuteroformamide was purchased from Eurisotop and used without further purification.
DCONH2. Trideuterated formamide (3.4 g, 71 mmol) and water (5 ml, 278 mmol) were mixed together and stirred for five minutes at room temperature. The mixture was evacuated (0.1 mbar) for one hour to remove water. The reaction was repeated three times. The formamide was purified by distillation (bp0.1 mm 60 °C). Yield 85% (2.77 g, 60 mmol).
N-deuteroformamide. Formamide (5.0 g, 111 mmol) and deuterated water (2.22 g, 111 mmol) were mixed together and stirred for five minutes at room temperature. The N-deuteroformamide was purified by distillation (bp0.1 mm 60 °C). Yield 95% with a 50% isotopic purity.
2.2. Spectroscopy
The measurements of rotational spectrum of deuterated formamide were performed in the frequency range 150–950 GHz using the Lille solid state (Motiyenko et al. 2010) and fast-scan (Alekseev et al. 2012) spectrometers. The large part of the frequency range was covered by solid state sources, and a BWO-based fast-scan spectrometer was used only in the frequency range 580–660 GHz. The measurement uncertainty is estimated to be 0.03 MHz for a single isolated line and 0.05 or 0.1 MHz for the lines with poor signal-to-noise ratio or distorted lineshape.
A part of the table available in CDS, with assigned rotational transitions of the ground states of deuterated isotopologues of formamide.
3. Results
Formamide is a prolate asymmetric top molecule (κ = 0.95). Therefore the Watson A-reduction Hamiltonian in Ir coordinate representation was chosen for modelling its rotational spectrum. In predicting the intensities of the spectra of deuterated species we used the dipole moment components from the parent one assuming that their variation is small upon isotopic substitution. The deuteration on amino group leads to two different molecular conformations. They are distinguished by the position of the carbonyl group hydrogen relative to the position of non-substituted hydrogen atom of the amino group, which could be either trans or cis.
The spectra of two samples whose synthesis is described above were studied. The first one contained almost pure DCONH2 species, and the second one contained a mixture of amino-deuterated species. As was deduced from the observed spectra, the relative abundance of the parent: cis-NHD: trans-NHD:ND2 isotopologues in the second sample can be approximately estimated as 46:22:22:10.
To simplify the initial assignment, the first frequency predictions of each deuterated species were calculated using the corresponding rotational rigid rotor constants A, B, C from (Costain & Dowling 1960) and the quartic centrifugal distortion constants from parent species (Kryvda et al. 2009). Such a combination of parameters was good enough to assign low-J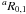 transitions in 150–200 GHz frequency range. The predicted line positions differed from the experimental ones by a few ten MHz. The following assignment was performed in a classical manner when the frequency predictions were improved step by step as new assigned lines were added to the fit. The nuclear quadrupole hyperfine structure was partially resolved for low-J transitions of all studied isotopomers, thus enabling determination of diagonal components of the nuclear quadrupole coupling tensor. For the lines with unresolved hyperfine structure, all hyperfine components were assigned to the same frequency and were fitted taking their relative intensities into account.
transitions in 150–200 GHz frequency range. The predicted line positions differed from the experimental ones by a few ten MHz. The following assignment was performed in a classical manner when the frequency predictions were improved step by step as new assigned lines were added to the fit. The nuclear quadrupole hyperfine structure was partially resolved for low-J transitions of all studied isotopomers, thus enabling determination of diagonal components of the nuclear quadrupole coupling tensor. For the lines with unresolved hyperfine structure, all hyperfine components were assigned to the same frequency and were fitted taking their relative intensities into account.
The results of the fits are presented in Table 1, which contains the values of fitted parameters and their standard deviations, number of lines in each dataset, rms, and weighted rms deviation of the fits, as well as maximum values of J and Ka numbers for assigned rotational lines. Thanks to the purity of the sample, hence more intense spectra, the number of lines assigned for DCONH2 is considerably greater than for all the other studied deuterated species of formamide. Both μa and μb transitions were assigned for all deuterated isotopologues. In particular, the assignment of μb transitions allowed an accurate determination of A and ΔK parameters. In general, for all these isotopologues we determined the full sets of quartic centrifugal distortion constants, as well as most of the sextic ones. All the datasets were fitted within the experimental accuracy as it follows from the values of weighted rms deviation we obtained. The fitted datasets include newly measured lines, as well as the measurements reported in earlier studies (Kurland & Wilson 1957; Costain & Dowling 1960). The measurement accuracy of the old studies has not been reported, but we estimate it to be about 0.1 MHz since almost all the residuals of the fit for the lines from Kurland & Wilson (1957) and Costain & Dowling (1960) are smaller than 0.1 MHz. However, several lines were excluded from the final fits because their residuals were higher than 1 MHz. The complete list of measured rotational transitions of the ground states of all studied deuterated isotopologues of formamide is presented in Table 2 available at the CDS. Here only a part of the Table 2 is shown as an example.
 |
Fig. 1 Predicted stick spectrum of the studied singly deuterated species of formamide at 150 K. The grey rectangle covers the frequency range of the direct measurements for the rotational spectrum in this study. |
4. Conclusions
The new extensive analysis of the rotational spectra of the ground vibrational states of all singly deuterated and –ND2 species of formamide was performed in the frequency range up to 1 THz. This represents significant improvement over the previously available results that only allowed for feasible predictions of the spectra of the studied isotopic species up to 40−50 GHz and for low values of J quantum number. The simulated spectra of singly deuterated species of formamide at the temperature of 150 K are represented in Fig. 1. As can be seen the new measurements cover the frequency range where the most intense lines occur. Therefore, these data can now be directly used for detecting deuterated formamide by, for example, the ALMA telescope, which will be fully operational in the next years. In addition, the sets of rotational and centrifugal distortion parameters obtained in this study of deuterated isotopologues can be used to accurately extrapolate their rotational spectra above 1 THz, at least for transitions involving quantum numbers J < 65 and Ka < 25. The frequencies predicted for transitions with higher numbers of J and Ka should be used with caution.
Acknowledgments
This work is supported by ANR-08-BLAN-0225, the French Programme National de Physique Chimie du Milieu Interstellaire, and by CNES. A.K. would like to acknowledge the support of the Embassy of France in Ukraine
References
- Alekseev, E. A., Motiyenko, R. A., & Margulès, L. 2012, Radio Physics and Radio Astronomy, 3, 75 [NASA ADS] [CrossRef] [Google Scholar]
- Bossa, J. B., Theule, P., Duvernay, F., & Chiavassa, T. 2009, ApJ, 707, 1524 [NASA ADS] [CrossRef] [Google Scholar]
- Charnley, S. B., Tielens, A. G. G. M., & Rodgers, S. D. 1997, ApJ, 482, L203 [NASA ADS] [CrossRef] [Google Scholar]
- Costain, C. C., & Dowling, J. M. 1960, J. Chem. Phys., 32, 158 [NASA ADS] [CrossRef] [Google Scholar]
- Garrod, R. T., Widicus Weaver, S. L., & Herbst, E. 2008, ApJ, 682, 283 [NASA ADS] [CrossRef] [Google Scholar]
- Halfen, D. T., Ilyushin, V., & Ziurys, L. M. 2011, ApJ, 743, 60 [NASA ADS] [CrossRef] [Google Scholar]
- Hollis, J. M., Lovas, F. J., Remijan, A. J., et al., 2006, ApJ, 643, L25 [Google Scholar]
- Jones, B. M., Bennett, C. J., & Kaiser, R. I. 2011, ApJ, 734, 78 [NASA ADS] [CrossRef] [Google Scholar]
- Kryvda, A. V., Gerasimov, V. G., Dyubko, S. F., et al. 2009, J. Mol. Spectrosc., 254, 28 [NASA ADS] [CrossRef] [Google Scholar]
- Kurland, R. J., & Wilson, E. B. Jr. 1957, J. Chem. Phys., 27, 585 [NASA ADS] [CrossRef] [Google Scholar]
- Motiyenko, R. A., Margulès, L., Alekseev, E. A., et al. 2010, J. Mol. Spectrosc., 264, 94 [Google Scholar]
- Motiyenko, R. A., Tercero, B., Cernicharo, J., et al. 2012, A&A, 548, A71 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Nagaoka, A., Watanabe, N., & Kouchi, A. 2005, ApJ, 624, L29 [NASA ADS] [CrossRef] [Google Scholar]
- Parise, B., Ceccarelli, C., Tielens, A. G. G. M., et al. 2006, A&A, 453, 949 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Ratajczak, A., Quirico, E., Faure, A., et al. 2009, A&A, 496, L21 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Ratajczak, A., Taquet, V., Kahane, C., et al. 2011, A&A, 528, L13 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Raunier, S., Chiavassa, T., Duvernay, F., et al. 2004, A&A, 416, 165 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Rubin, R. H., Swenson, G. W., Benson, R. C., et al., 1971, ApJ, 169, L39 [NASA ADS] [CrossRef] [Google Scholar]
- Turner, B. E. 1991, ApJSS, 76, 617 [CrossRef] [Google Scholar]
- Woodall, J., Agundez, M., Markwick-Kemper, A. J., & Millar, T. J. 2007, A&A, 466, 1197 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
All Tables
A part of the table available in CDS, with assigned rotational transitions of the ground states of deuterated isotopologues of formamide.
All Figures
 |
Fig. 1 Predicted stick spectrum of the studied singly deuterated species of formamide at 150 K. The grey rectangle covers the frequency range of the direct measurements for the rotational spectrum in this study. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


