| Issue |
A&A
Volume 546, October 2012
|
|
|---|---|---|
| Article Number | A81 | |
| Number of page(s) | 6 | |
| Section | Planets and planetary systems | |
| DOI | https://doi.org/10.1051/0004-6361/201219886 | |
| Published online | 09 October 2012 | |
Implantation of multiply charged carbon ions in water ice
1
Centre de Recherche sur les Ions, les Matériaux et la Photonique
(CEA/CNRS/ENSICAEN/UCBN), CIMAP – CIRIL – GANIL, Boulevard Henri Becquerel, BP 5133
14070, Caen Cedex 05, France
e-mail: This email address is being protected from spambots. You need JavaScript enabled to view it.
2
School of Nuclear Science and Technology, Lanzhou
University, 730000
Lanzhou, PR
China
3
Departamento de Disciplinas Básicas e Gerais,
CEFET-RJ, Av. Maracanã 229,
20271-110
Rio de Janeiro, RJ, Brazil
4
Departamento de Física, Pontifícia Universidade Católica do Rio de
Janeiro, Rua Marquês de São Vicente
225, 22451-900,
Rio de Janeiro, RJ, Brazil
5
Laboratory for Atomic and Surface Physics, University of
Virginia, 351 McCormick
Road, PO Box
400238, Charlottesville, VA
22904-4238,
USA
6
Centre for Plasma Physics, School of Mathematics and Physics,
Queen’s University, Belfast, Belfast BT7
1NN, Northern Ireland
7
Department of Physics and Astronomy, The Open
University, Walton
Hall, Milton
Keynes, UK
8
INAF Osservatorio Astrofisico di Catania,
Catania,
Italy
9
IPD/UNIVAP, Av. Shishima Hifumi, 2911 São Jose dos Campos, SP, Brazil
Received: 25 June 2012
Accepted: 28 July 2012
Abstract
Context. Several objects in the solar system like Europa, Ganymede, and Callisto have frozen surface (main component: H2O). The associated thickness is bigger than the penetration depth of the relevant projectile ions. Additionally, other species such as H2O2, SO2, and CO2 have been detected on these surface. The formation mechanisms of these molecules are still under discussion.
Aims. We present new experimental results on the implantation of 13Cq+ (q = 2, 3) ions at an energy of 30 keV in water ice at low temperatures (15 and 80 K). Experiments with multiply-charged ions at energies of tens of keV are particularly relevant to simulating the complexity of the irradiation environment to which the surfaces of the icy moons in the outer solar system are exposed.
Methods. The experiments were performed at the low-energy ion beam facility ARIBE of GANIL in Caen (France). 30 keV 13Cq+ (q = 2, 3) ions have been used to bombard solid H2O surface, which were frozen at 15 K and 80 K. Fourier transform infrared spectrometer (FTIR) was used to analyze the sample in the 5000–600 cm-1 (2–16.7 μm) region with a spectral resolution of 1 cm-1.
Results. The results of our experiments indicate that implantation produces 13CO2 with yields in the range of 0.32–0.57 molecules ion-1. This yield seems to be independent of the temperature of the ices in the range studied. We have estimated the time scale necessary to accumulate by implantation of magnetospheric carbon ions the observed quantity of carbon dioxide on the surface of Europa, a Jovian moon. This time scale is of the order of 1.0–1.3 × 104 yrs, which is higher than that evaluated for carbon dioxide production by other relevant processes.
Conclusions. We conclude that although a relevant quantity of CO2 can be formed by carbon ion implantation, this is not the dominant formation mechanism at Europa.
Key words: planets and satellites: surfaces / methods: laboratory / techniques: spectroscopic
© ESO, 2012
1. Introduction
Ion implantation is a very common technique for the doping (i.e. introduction of impurities) of different materials. It is widely used in the semiconductor industries for e.g. modifying the electrical properties (such as the threshold voltage) of transistors. Dopant ions are produced, accelerated, selected by the mass-to-charge ratio, and directed to the target. The ions penetrate into the target, collide with the host atoms, lose energy, and remain implanted at some depth within the solid. The penetration depth depends on the mass and energy of the incoming ion and on the properties of the target. Experiments are performed with energies from several hundred to several million electron volts, resulting in ion implantation depths from <10 nm to >10 μm.
Implantation experiments are also relevant in very different scenarios as those of reactive ions (H, O, C, N, S; Baratta et al. 2012) irradiating ices in several astrophysical environments. Implanted ions deposit energy in the icy target (for a detailed description of the phenomenology of interaction between fast ions and target species see Johnson 1990). In addition only reactive ions (carbon in this paper) have a chance to form species that include the implanted projectile with a maximum yield of one molecule per incoming ion.
There are several objects in the solar system where ices on planetary surfaces, comets, etc., are much thicker than the penetration depth of the relevant ion populations. As an example, earth based and space observations indicate that on the surface of Europa, Ganymede, and Callisto water ice is the dominant species along with hydrated materials and minor amounts of some volatile species such as H2O2, SO2 and CO2 (Carlson et al. 1997, 1999, 2001; Noll et al. 1995, 1997). The formation mechanism of those molecules is still an open question. A possible way is via exogenic processes such as implantation of carbon or sulfur ions of the Jovian magnetosphere, as suggested since several years on the basis of numerous experimental results (for a review see Strazzulla 2011).
In this paper we present new experimental results on the implantation of 13Cq+ (q = 2, 3) ions at an energy of 30 keV in water ice at low temperatures (15 and 80 K). Experiments with multiply charged ions at energies of tens of keV are particularly relevant to simulating the complexity of the bombardment environment to which the surfaces of the icy moons in the outer solar system are exposed.
The present results confirm and extend those obtained by bombardment of frozen layers of pure H2O ice with 10–30 keV singly charged (Bibring & Rocard 1984; Strazzulla et al. 2003) and singly/doubly charged 2–4 keV (Dawes et al. 2007; Hunniford et al. 2009) carbon ions. The results of our experiments indicate that implantation produces 13CO2 with yields in the range of 0.32–0.57 molecules ion-1.
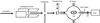 |
Fig. 1 Schematic diagram of our experimental apparatus. |
Summary of implantation yields in molecules ion-1.
2. Experiments
The experiments were performed at the low energy ion beam facility ARIBE of GANIL in Caen (France). 13Cq+ (q = 2, 3) ion beams (~1012 ions cm-2 s-1) were produced in a 14.5 GHz electron cyclotron resonance (ECR) ion source. After selection of mass and charge by a dipole magnet, the beams were guided toward the experimental set-up (shown in Fig. 1). A sweeping device made of two parallel plates assured a uniform bombardment of the target. The beam passed through an insulated collimator. A part of the beam was stopped by the collimator and the corresponding collimator current was measured. A Faraday-Cup was inserted into the beam about 12 cm upstream of the target. From the ratio of the current measured on the collimator and in the Faraday-Cup, the beam flux (number of projectiles per square centimeter and second) on the target can be determined. The accumulated charge on the collimator was measured by means of current integrators. From this accumulated charge and the ratio of the current on the collimator and the current in the Faraday-Cup, the projectile fluence (number of projectiles accumulated per square centimeter) could be determined. The ratio was checked several times before, during and after bombardment to monitor for any variation of the flux.
 |
Fig. 2 Spectra of water ices bombarded with 30 keV 13C3+ and 13C2+ ions at 15 K and 80 K, respectively. Dashed lines represent spectra before bombardment, solid lines represent spectra after bombardment (about 1016 ions cm-2). |
The ice layers were prepared by condensing water in the gas phase on a CsI window at 15 K and 80 K. About 1 cm3 pure H2O which was produced by a Milli-Q Integral Water Purification System was stored in a cuvette. The H2O was frozen to solid phase, then while pumping the cuvette for a few minutes, the H2O was warmed up to liquid phase. This procedure was repeated 2 or 3 times in order to obtain of H2O vapor of high purity. H2O vapor was introduced in a pre-chamber. A fine valve, which allowed controlling the deposition rate, was used to transmit the gas into the high vacuum chamber and onto the cold CsI substrate. The pressure in the high vacuum chamber was below 10-7 mbar during the measurements. The CsI window was installed in the center of the chamber on a cold finger connected to a closed cycle helium cryostat. The temperature of the substrate was controlled by a carbon resistance and a compound linear thermal sensor (CLTS) situated on the holder, providing a precision of 0.1 K. The cold head with the CsI window could be rotated from 0° to 180° and fixed in three positions allowing bombardment (0°), FTIR analysis (90°) and deposition (180°). A Nicolet Magna 550 Fourier Transform Infrared Spectrometer (FTIR) was used to analyze the sample in the 5000–600 cm-1 (2–16.7 μm) region with a spectral resolution of 1 cm-1. The spectra were always corrected by a background recorded before deposition.
The thickness of the sample (estimated from their column density assuming an ice density of 1 g cm-3, details of calculation can be found in Pilling et al. 2011) was between 0.7 μm and 1.8 μm (Table 1). The range (or penetration depth) of 30 keV carbon ions in water ice is of the order of 178 ± 50 nm (Ziegler et al. 2008). Thus, the ices that we deposited were sufficiently thick to assure complete stopping and implantation of all projectiles.
3. Results
Figure 2 shows spectra measured before and after bombardment of H2O ices deposited at 15 K and 80 K, respectively, with 30 keV 13Cq+ (q = 2, 3) ions. Up to two different absorption bands are observed. The band at 2346 cm-1 belongs to the ν3 vibration mode of 12CO2 (Sandford & Allamandola 1990; Palumbo et al. 1998; Jamieson et al. 2006; Seperuelo Duarte et al. 2010; Pilling et al. 2010, 2011) and is observed before bombardment. We believe that such an impurity mainly comes from residual gas in the bombardment chamber but not from CO2 impurities in the original water sample. Indeed the column density of 12CO2 increases during the experiment due to additional deposition from residual gas. Probably 12CO2 is adsorbed on some of the walls and released because of the interaction with the ion beam that is sweeped on a large area to produce an uniform implantation.
The band at 2277 cm-1 belongs to the ν3 vibration mode of 13CO2 (Gerakines et al. 1995; Strazzulla et al. 2005; Seperuelo Duarte et al. 2010). The intensity of this band is increasing during the bombardment, indicating that the corresponding new formed 13CO2 molecules are clearly correlated with the implantation of 13Cq+. The chemical processes which start with dissociation of H2O molecule and take place in the ice are: 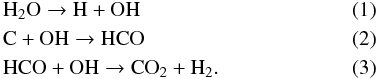 As mentioned by Lv et al. (2011), 13CO have not detected after carbon implantation into pure H2O ices, both at 15 K and 80 K. 13CO can be produced by dissociation of 13CO2 at higher dose when 13CO2 is abundant (see Strazzulla et al. 2003). The chemistry changes if the implanted target contains O2. In that case CO is formed first (Boduch et al. 2012).
As mentioned by Lv et al. (2011), 13CO have not detected after carbon implantation into pure H2O ices, both at 15 K and 80 K. 13CO can be produced by dissociation of 13CO2 at higher dose when 13CO2 is abundant (see Strazzulla et al. 2003). The chemistry changes if the implanted target contains O2. In that case CO is formed first (Boduch et al. 2012).
We calculated the column density of 12CO2 and 13CO2 by assuming a band strength of 7.6 × 10-17 cm molecule-1 and 7.8 × 10-17 cm molecule-1, respectively (Yamada & Person 1964; Gerakines et al. 1995; Jamieson et al. 2006). This was accomplished by using Eq. (4): 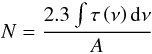 (4)where N represents the column density (in molecules cm-2) of 12CO2 and 13CO2, A represents the band strength (in cm molecule-1), ∫τ(ν)dν represents the area (in cm-1) of a given measured band on absorbance scale. On the one hand, 13CO2 is produced in the target by implantation of 13Cq+ ions. On the other hand, 12CO2 from the residual gas could also condense on the substrate. This has been taken into account by assuming a natural isotopic ratio of 12C and 13C in the atmosphere of about 90 to determine the amount of 13CO2 associated with the implantation process.
(4)where N represents the column density (in molecules cm-2) of 12CO2 and 13CO2, A represents the band strength (in cm molecule-1), ∫τ(ν)dν represents the area (in cm-1) of a given measured band on absorbance scale. On the one hand, 13CO2 is produced in the target by implantation of 13Cq+ ions. On the other hand, 12CO2 from the residual gas could also condense on the substrate. This has been taken into account by assuming a natural isotopic ratio of 12C and 13C in the atmosphere of about 90 to determine the amount of 13CO2 associated with the implantation process.
Figure 3 shows the column density of produced 13CO2 as a function of the fluence of implanted 30 keV 13C3+ and 13C2+ ions into water ices at 15 K and 80 K. Also included are experimental data obtained in Catania (Italy), for 30 keV 13C+ implantation in water ice at slightly different temperatures of 16 K and 77 K (Strazzulla et al. 2003), for comparison. 13CO2 production yields are shown in the fifth column of Table 1. Within error bars, which stem from the uncertainties of the determination of the column density on one hand, and the uncertainty of the fluence measurement on the other hand, the results obtained in Catania for 13C+ and in Caen for 13C2+ and 13C3+ are in good agreement. All of these results can be summarized as follows:
-
13CO2 was produced after implantation of 30 keV 13Cq+ ions with different charge states into H2O ices deposited at different temperatures.
-
No effects related to the temperature of the ices have been observed.
-
No effects related to the charge states of the incident ions have been found.
The main difference between samples deposited at 15 K and 80 K is that the sample deposited at lower temperature has a higher porosity than that deposited at higher temperature (Baragiola 2003; Palumbo 2006). This has been confirmed by the observation, in our spectra, of the O–H dangling bonds (db) bands that peak at about 3720 and 3695 cm-1 in water ice deposited at 15 K and at 3691 cm-1 for 80 K ice (Palumbo & Strazzulla 2003; Palumbo 2005). Pilling et al. (2011) have shown that for H2O:CO2 (1:1) ices, the sample deposited at 80 K has the OH db (3690–3633 cm-1) area about 10–50 times smaller than the sample deposited at 13 K. Our result that no temperature effects have been observed shows that the porosity does not play an important role in the formation of carbon dioxide during 30 keV 13Cq+ interaction with pure water ices.
 |
Fig. 3 Column density of 13CO2 produced after implantation of 30 keV 13Cq+ (q = 1, 2, 3) ions at 15 K and 80 K. The experimental data for 13C+ implanted at 16 K and 77 K in water ice were obtained in Catania (Strazzulla et al. 2003). The solid lines represent linear least square fits, the corresponding production yields are shown in Table 1. |
The interaction of slow ions impacting on solid surface has been studied extensively (Arnau et al. 1997). Ions approaching the surface will capture electrons from the target surface to the excited states of the ions (the number of captured electrons depending on the charge states of the incoming ions). These electron capture processes take place only at the surface. As soon as the projectiles enter into the bulk, they will rapidly reach the effective charge state (typically within one single monolayer) which should be the same for all of the implanted 13C2+ and 13C3+ ions at the same velocity (Herrmann et al. 1994). The memory of the incoming charge state is lost. Indeed, our results show that the values of the 13CO2 production yields do not vary with the projectile charge state. The mean value is 0.45 molecules of 13CO2 formed per incoming projectile ion.
Dawes et al. (2007) reported results on 4 keV 13C+ and 13C2+ implantation into water ice deposited at 30 K and 90 K (shown in Fig. 4). For 13C+ ions, the column density of synthesized 13CO2 showed an asymptotic increase, reaching saturation at higher fluences. For 13C2+ ions, the evolution of the column density of synthesized 13CO2 with fluence can be described as the sum of two asymptotically increasing functions (see also Hunniford et al. 2009). Viewing the result as a whole (shown in Fig. 4 where also some of our results are reported to facilitate the comparison), the reported yields of 13CO2 by Dawes et al. (2007) are much smaller than those observed in the experiments at both Catania (Strazzulla et al. 2003) and Caen (this work, shown in Table 1).
 |
Fig. 4 Comparsion of 13CO2 the production yields between our results and the results of Dawes et al. (2007). Data from singly and doubly charged projectiles are presented in left and right figures, respectively. |
The electronic stopping power of 30 keV 13Cq+ (Se ≈ 85 keV μm-1) dominates over the nuclear stopping power (Sn ≈ 62 keV μm-1). For 4 keV 13Cq+ ions, the nuclear stopping power (Sn ≈ 110 keV μm-1) is much larger than the electronic stopping power (Se ≈ 30 keV μm-1) that we calculated by using the available software SRIM-2008 (Ziegler et al. 2008). Comparing our results to those of Dawes et al. (2007) one could be led to the conclusion that electronic stopping power plays an important role in driving chemical reactions between implanted particles and neighbor radicals. According to a SRIM 2008 calculation (Ziegler et al. 2008), the sputtering yields were found to be 0.15 and 0.33 molecules ion-1 for 30 keV 13Cq+ and 4 keV 13Cq+ ions respectively. This means that at the end of the bombardment, only less than 10 monolayers H2O molecules have been sputtered by 4 keV carbon ions, while the number of molecules sputtered by 30 keV ions can be neglected. This is not sufficient to explain the differences of 13CO2 production yield between 30 keV and 4 keV 13Cq+ ions. The amount of ions associated with the back scattering effect is less than 1% for both projectiles, therefore this effect can also be neglected. The penetration depth were found to be of the order of 178 ± 50 and 25 ± 9 nm for 30 keV 13Cq+ and 4 keV 13Cq+ ions, respectively (Ziegler et al. 2008). This means that 30 keV 13Cq+ ions interact with a greater number of H2O molecules than 4 keV 13Cq+ ions. If the implantation yields are compared to the number of H2O molecules exposed to the ions, the ratio between implantation yields of 30 keV 13Cq+ and 4 keV 13Cq+ ions should be around 7 to 1, but this is not that case in our experimental results. Also, a larger longitudinal and lateral straggling is more important for 30 keV 13Cq+ ions. Since the sputtering effect and the amount of back scattered ions are negligible, only the range of the scattering region could explain the difference of our present results with those of Dawes et al. (2007). This difference, however remains an open question. Complementary experiments are needed to understand the basic physical processes occurring during implantation.
4. Discussion and conclusion
The surfaces of the icy moons in the Jovian system are subjected to intense bombardment by electrons, protons and multiply charged ions (Johnson et al. 2004; Bagenal et al. 2007). It is usual to refer to ions and electrons at or below 10 keV as “plasma” and above as “energetic particles”. The plasma nearly corotates with Jupiter and then it flows preferentially onto the hemisphere trailing the satellite’s motion. The less abundant but higher energy particles bombard the satellite in more complex ways exhibiting different spatial distributions depending on their energy, mass, charge, and the electric and magnetic fields near each moon (see Johnson et al. 2004, and references therein).
The temperatures of the water ice samples investigated in this paper are adequate to simulate those measured on many of the icy moons in the external solar system. As examples the temperatures of the Europa’ surface are measured to be between 50 and 125 K; on Enceladus they are between 30 and 145 K and between 50 and 100 K on Rhea (McFadden et al. 2007; Spencer et al. 2006).
With this in mind it is clear that measuring in the laboratory parameters such as the implantation yields of relevant ions (C in the present case) in a wide range of charge states and energies are of great interest. Nowadays the progress in the field of space observations yields spatial resolutions high enough that the amount of a given chemical species can be mapped on the surface and related to exogenic effects such as magnetospheric charged particle bombardment, ultraviolet photolysis, impacts. As an example analysis of data relative to Europa have shown that sulfuric acid hydrate abundance is independent of underlying geologic unit, but is very well correlated with the flux of bombarding sulfur ions (Dalton et al. 2011). Interestingly a first set of experiments have demonstrated that sulfur ion implantation produces hydrated sulfuric acid with a high yield (Strazzulla et al. 2007).
At present CO2 has been observed on the surfaces of the Galilean moons and other objects in the outer solar system (for a review see Dalton et al. 2010). Its abundance and its superficial distribution is not known. McCord et al. (1997, 1998) first noticed that CO2, because of the absence of vibro-rotational spectral signatures, should be in a condensed phase, possibly dispersed in another material. The same authors evaluated that a CO2 column density as low as 3 × 1017 molecules cm-2 is required to produce the absorption observed at Europa McCord et al. (1997, 1998).
Different possibilities exist to explain its presence at the surface. Some are endogenic: it could be degassed from internal sources (Moore et al. 1999). Other mechanisms are exogenic such as those that have to do with energetic processing of the surface by ions, electrons and photons and/or with carbon implantation. Concerning the energetic processing by ions, a large amount of experiments have shown that icy mixtures of H2O with a plethora of carbon-bearing molecules (e.g. CO, CH4, CH3OH etc.) all produce CO2 although with different efficiencies (Palumbo et al. 1998; Ioppolo et al. 2009; Pilling et al. 2012). The same is also true for photons and electrons (Watanabe et al. 2007; Baratta et al. 2002; Gerakines et al. 1996; Wada et al. 2006; Bennett et al. 2011). It is also been demonstrated that CO2 can be formed at the interface between water ice (Mennella et al. 2004; Gomis & Strazzulla 2005; Raut et al. 2012) or O2 (Fulvio et al. 2012) and it has been suggested that this could produce enough carbon dioxide to explain the observed amount.
Concerning the possibility that carbon implantation plays an important role, the knowledge of the flux of carbon ions impinging on the surfaces of the moons is needed. Those fluxes are, however, not known with great precision. A rough estimation of the average flux of carbon ions has been used by Strazzulla et al. (2003) In fact the fluxes of sulphur (keV–MeV) ions at the surfaces of the galilean moons have been reported (it values ~9 × 106 S ions cm-2 s-1 at Europa) (Cooper et al. 2001) and although the abundance of carbon ions has not been quantified, the number ratio C/S has been reported. As an example, it amounts to about 0.2 near by Europa in the 1 MeV nucl-1 range (Hamilton et al. 1981; Cohen et al. 2001). Assuming C/S = 0.2 at Europa, and using the experimental values ranging between 0.4 and 0.5 CO2 molecules (impinging-ion)-1 as production yield (see table 1; here we use the results obtained at 80 K, a temperature more appropriate to the Jovian moons) we can evaluate the time necessary to produce a column density of 3 × 1017 molecules cm-2. This time scale results to be, at Europa, on the order of 1.0–1.3 × 104 yrs. This time has to be compared with those of other processes. It turns out that the estimated time scale for the production of carbon dioxide by ion bombardment of water on top of carbonaceous materials ranges between 50 and 2 × 103 yrs (Gomis & Strazzulla 2005). We conclude that although a relevant quantity of CO2 can be formed by carbon ion implantation, this is not the dominant formation mechanism.
It is trivial to say that more observational data on carbon ion flux distribution at the surfaces of icy moons are needed as well as their relationship with the local CO2 abundance.
Acknowledgments
This research has been partially supported by the European COST Action CM 0805: The Chemical Cosmos: Understanding Chemistry in Astronomical Environments. We acknowledge CAPES, COFECUB, the Basse-Normandie region for financial support. We thank S. Guillous, T. Been, F. Noury and J. M. Ramillon for their technical assistance.
References
- Arnau, A., Aumayr, F., Echenique, P. M., et al. 1997, Surf. Sci. Rep., 27, 113 [Google Scholar]
- Bagenal, F., Dowling, T. E., & McKinnon, W. B. 2007, Jupiter: the planet, satellites and magnetosphere (Cambridge Univ. Pr.), 1 [Google Scholar]
- Baragiola, R. A. 2003, Planet. Space. Sci., 51, 953 [NASA ADS] [CrossRef] [Google Scholar]
- Baratta, G. A., Leto, G., & Palumbo, M. E. 2002, A&A, 384, 343 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Baratta, G. A., Fulvio, D., Garozzo, M., et al. 2012, Mem. Soc. Astron. It. Supp., 20, 94 [Google Scholar]
- Bennett, C., Hama, T., Kim, Y., Kawasaki, M., & Kaiser, R. 2011, ApJ, 727, 27 [NASA ADS] [CrossRef] [Google Scholar]
- Bibring, J. P., & Rocard, F. 1984, Adv. Space Res., 4, 103 [NASA ADS] [CrossRef] [Google Scholar]
- Boduch, P., Domaracka, A., Fulvio, D., et al. 2012, A&A, 544, A30 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Carlson, R. W. 2001, in Division for Planetary Sciences Meeting Abstracts, 33, 1125 [Google Scholar]
- Carlson, R. W., Smythe, W. D., Lopes-Gautier, R. M. C., et al. 1997, Geophys. Res. Lett., 24, 2479 [NASA ADS] [CrossRef] [Google Scholar]
- Carlson, R. W., Anderson, M. S., Johnson, R. E., et al. 1999, Science, 283, 2062 [NASA ADS] [CrossRef] [PubMed] [Google Scholar]
- Cohen, C. M. S., Stone, E. C., & Selesnick, R. S. 2001, J. Geophys. Res., 106, 29871 [NASA ADS] [CrossRef] [Google Scholar]
- Cooper, J. F., Johnson, R. E., Mauk, B. H., Garrett, H. B., & Gehrels, N. 2001, Icarus, 149, 133 [NASA ADS] [CrossRef] [Google Scholar]
- Dalton, J. B., Cruikshank, D. P., Stephan, K., et al. 2010, Space. Sci. Rev., 153, 113 [Google Scholar]
- Dalton, J. B., Shirley, J. H., Cassidy, T., Paranicas, C. J., & Kamp, L. W. 2011, in EPSC-DPS Joint Meeting, 649 [Google Scholar]
- Dawes, A., Hunniford, A., Holtom, P. D., et al. 2007, PCCP, 9, 2886 [Google Scholar]
- Fulvio, D., Raut, U., & Baragiola, R. A. 2012, ApJ, 752, L33 [NASA ADS] [CrossRef] [Google Scholar]
- Gerakines, P. A., Schutte, W. A., Greenberg, J. M., & van Dishoeck, E. F. 1995, A&A, 296, 810 [NASA ADS] [Google Scholar]
- Gerakines, P., Schutte, W., & Ehrenfreund, P. 1996, A&A, 312, 289 [NASA ADS] [Google Scholar]
- Gomis, O., & Strazzulla, G. 2005, Icarus, 177, 570 [NASA ADS] [CrossRef] [Google Scholar]
- Hamilton, D. C., Gloeckler, G., Krimigis, S. M., & Lanzerotti, L. J. 1981, J. Geophys. Res., 86, 8301 [NASA ADS] [CrossRef] [Google Scholar]
- Herrmann, R., Cocke, C. L., Ullrich, J., et al. 1994, Phys. Rev. A, 50, 1435 [NASA ADS] [CrossRef] [Google Scholar]
- Hunniford, C. A., Dawes, A., Fulvio, D., et al. 2009, in J. Phys.: Conf. Ser. (IOP Publishing), 163, 012078 [Google Scholar]
- Ioppolo, S., Palumbo, M. E., Baratta, G. A., & Mennella, V. 2009, A&A, 493, 1017 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Jamieson, C. S., Mebel, A. M., & Kaiser, R. I. 2006, ApJS., 163, 184 [NASA ADS] [CrossRef] [Google Scholar]
- Johnson, R. E. 1990, Energetic charged-particle interactions with atmospheres and surfaces (Heidelberg: Springer-Verlag), 19 [Google Scholar]
- Johnson, R. E., Carlson, R. W., Cooper, J. F., et al. 2004, Jupiter: The Planet, Satellites and Magnetosphere, 485 [Google Scholar]
- Lv, X. Y., Boduch, P., Bordalo, V., et al. 2011, in EPSC-DPS Joint Meeting 2011, 299 [Google Scholar]
- McCord, T. B., Carlson, R. W., Smythe, W. D., et al. 1997, Science, 278, 271 [NASA ADS] [CrossRef] [PubMed] [Google Scholar]
- McCord, T. B., Hansen, G. B., Clark, R. N., et al. 1998, J. Geophys. Res., 103, 8603 [NASA ADS] [CrossRef] [Google Scholar]
- McFadden, L. A. A., Weissman, P. R., & Johnson, T. V. 2007, Encyclopedia of the solar system (Academic) [Google Scholar]
- Mennella, V., Palumbo, M. E., & Baratta, G. A. 2004, ApJ, 615, 1073 [NASA ADS] [CrossRef] [Google Scholar]
- Moore, J. M., Asphaug, E., Morrison, D., et al. 1999, Icarus, 140, 294 [Google Scholar]
- Noll, K. S., Weaver, H. A., & Gonnella, A. M. 1995, J. Geophys. Res., 100, 19 [Google Scholar]
- Noll, K. S., Johnson, R. E., McGrath, M. A., & Caldwell, J. J. 1997, Geophys. Res. Lett., 24, 1139 [NASA ADS] [CrossRef] [Google Scholar]
- Palumbo, M. E. 2005, in J. Phys.: Conf. Ser. (IOP Publishing), 6, 211 [Google Scholar]
- Palumbo, M. E. 2006, A&A, 453, 903 [NASA ADS] [CrossRef] [EDP Sciences] [MathSciNet] [Google Scholar]
- Palumbo, M. E., & Strazzulla, G. 2003, Can. J. Phys., 1, 217 [NASA ADS] [CrossRef] [Google Scholar]
- Palumbo, M. E., Baratta, G. A., Brucato, J. R., et al. 1998, A&A, 334, 247 [NASA ADS] [Google Scholar]
- Pilling, S., Seperuelo Duarte, E., Domaracka, A., et al. 2010, A&A, 523, A77 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Pilling, S., Duarte, E. S., Domaracka, A., et al. 2011, PCCP, 13, 15755 [NASA ADS] [CrossRef] [Google Scholar]
- Pilling, S., Andrade, D. P. P., da Silveira, E. F., et al. 2012, MNRAS, 423, 2209 [NASA ADS] [CrossRef] [Google Scholar]
- Raut, U., Fulvio, D., Loeffler, M. J., & Baragiola, R. A. 2012, ApJ, 752, 159 [NASA ADS] [CrossRef] [Google Scholar]
- Sandford, S. A., & Allamandola, L. J. 1990, Icarus, 87, 188 [NASA ADS] [CrossRef] [PubMed] [Google Scholar]
- Seperuelo Duarte, E., Domaracka, A., Boduch, P., et al. 2010, A&A, 512, A71 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Spencer, J. R., Pearl, J. C., Segura, M., et al. 2006, Science, 311, 1401 [NASA ADS] [CrossRef] [PubMed] [Google Scholar]
- Strazzulla, G. 2011, Nucl. Instrum. Meth. B, 269, 842 [Google Scholar]
- Strazzulla, G., Leto, G., Gomis, O., & Satorre, M. A. 2003, Icarus, 164, 163 [NASA ADS] [CrossRef] [Google Scholar]
- Strazzulla, G., Leto, G., Spinella, F., & Gomis, O. 2005, Astrobiology, 5, 612 [NASA ADS] [CrossRef] [PubMed] [Google Scholar]
- Strazzulla, G., Baratta, G. A., Leto, G., & Gomis, O. 2007, Icarus, 192, 623 [NASA ADS] [CrossRef] [Google Scholar]
- Wada, A., Mochizuki, N., & Hiraoka, K. 2006, ApJ, 644, 300 [NASA ADS] [CrossRef] [Google Scholar]
- Watanabe, N., Mouri, O., Nagaoka, A., et al. 2007, ApJ, 668, 1001 [NASA ADS] [CrossRef] [Google Scholar]
- Yamada, H., & Person, W. B. 1964, J. Chem. Phys., 41, 2478 [NASA ADS] [CrossRef] [Google Scholar]
- Ziegler, J. F., Biersack, J. P., & Ziegler, M. D. 2008, Stopping and range of ions in matter (SRIM Co.) [Google Scholar]
All Tables
All Figures
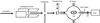 |
Fig. 1 Schematic diagram of our experimental apparatus. |
| In the text | |
 |
Fig. 2 Spectra of water ices bombarded with 30 keV 13C3+ and 13C2+ ions at 15 K and 80 K, respectively. Dashed lines represent spectra before bombardment, solid lines represent spectra after bombardment (about 1016 ions cm-2). |
| In the text | |
 |
Fig. 3 Column density of 13CO2 produced after implantation of 30 keV 13Cq+ (q = 1, 2, 3) ions at 15 K and 80 K. The experimental data for 13C+ implanted at 16 K and 77 K in water ice were obtained in Catania (Strazzulla et al. 2003). The solid lines represent linear least square fits, the corresponding production yields are shown in Table 1. |
| In the text | |
 |
Fig. 4 Comparsion of 13CO2 the production yields between our results and the results of Dawes et al. (2007). Data from singly and doubly charged projectiles are presented in left and right figures, respectively. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


