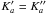| Issue |
A&A
Volume 541, May 2012
|
|
|---|---|---|
| Article Number | A121 | |
| Number of page(s) | 8 | |
| Section | Interstellar and circumstellar matter | |
| DOI | https://doi.org/10.1051/0004-6361/201118738 | |
| Published online | 11 May 2012 | |
The quest for complex molecules in space: laboratory spectroscopy of n-butyl cyanide, n-C4H9CN, in the millimeter wave region and its astronomical search in Sagittarius B2(N)⋆
1
I. Physikalisches Institut, Universität zu Köln,
Zülpicher Str. 77, 50937
Köln, Germany
e-mail: hspm@ph1.uni-koeln.de
2
Max-Planck Institut für Radioastronomie,
Auf dem Hügel 69, 53121
Bonn,
Germany
3
Université de Toulouse, UPS-OMP, IRAP, Toulouse, France
4
CNRS, IRAP, 9
Av. colonel Roche, BP
44346, 31028
Toulouse Cedex 4,
France
Received: 23 December 2011
Accepted: 8 March 2012
Context. The saturated n-propyl cyanide was recently detected in Sagittarius B2(N). The next largest unbranched alkyl cyanide is n-butyl cyanide.
Aims. We provide accurate rest frequency predictions beyond the millimeter wave range to search for this molecule in the Galactic center source Sagittarius B2(N) and facilitate its detection in space.
Methods. We investigated the laboratory rotational spectrum of n-butyl cyanide between 75 GHz and 348 GHz. We searched for emission lines produced by the molecule in our sensitive IRAM 30 m molecular line survey of Sagittarius B2(N).
Results. We identified more than one thousand rotational transitions in the laboratory for each of the three conformers for which limited data had been obtained previously in a molecular beam microwave study. The quantum number range was greatly extended to J ≈ 120 or more and Ka > 35, resulting in accurate spectroscopic parameters and accurate rest frequency calculations up to about 500 GHz for strong to moderately weak transitions of the two lower energy conformers. Upper limits to the column densities of N ≤ 3 × 1015 cm-2 and 8 × 1015 cm-2 were derived towards Sagittarius B2(N) for the two lower energy conformers, anti-anti and gauche-anti, respectively.
Conclusions. Our present data will be helpful for identifying n-butyl cyanide at millimeter or longer wavelengths with radio telescope arrays such as ALMA, NOEMA, or EVLA. In particular, its detection in Sagittarius B2(N) with ALMA seems feasible.
Key words: molecular data / methods: laboratory / techniques: spectroscopic / radio lines: ISM / ISM: molecules / ISM: individual objects:Sagittarius B2
Full Table 3 and Tables 4–5 are only available at the CDS via anonymous ftp to cdsarc.u-strasbg.fr (130.79.128.5) or via http://cdsarc.u-strasbg.fr/viz-bin/qcat?J/A+A/541/A121
© ESO, 2012
1. Introduction
Cyanides account for 20 of the approximately 165 molecules detected in the interstellar medium or in the circumstellar envelopes of late-type stars1; not included in this number are isocyanides and other molecules containing the CN moiety. Saturated cyanides are usually found in high-mass star-forming regions, as are other saturated molecules. One part of the Galactic center source Sagittarius (Sgr for short) B2(N) was nicknamed Large Molecule Heimat because of the many and in part rather complex molecules found there, many of them for the first time (Snyder et al. 1994; Menten 2004). Line surveys of the two hot cores Sgr B2(N) and Sgr B2(M) at 3 mm, with selected observations at higher frequencies, have been carried out with the IRAM 30 m telescope to investigate the molecular complexity in these prolific sources. Two cyanides, aminoacetonitrile (NH2CH2CN, Belloche et al. 2008), a potential precursor to glycine, and n-propyl cyanide along with ethyl formate (n-C3H7CN and C2H5OCHO, Belloche et al. 2009) were detected for the first time in space toward Sgr B2(N) in the course of this study. In addition, 13C isotopologs of vinyl cyanide (C2H3CN) were detected for the first time in the interstellar medium, and those of ethyl cyanide (C2H5CN) for the first time in this source (Müller et al. 2008); the latter had been identified shortly before in a line survey of Orion KL (Demyk et al. 2007).
With the recent detection of n-propyl cyanide, the series of unbranched alkyl cyanides detected in the interstellar medium now contains three members: methyl cyanide, ethyl cyanide, and n-propyl cyanide. The chemical models of Belloche et al. (2009) succeeded in reproducing the measured column density ratios with a sequential, piecewise construction of these alkyl cyanides from their constituent functional groups on the grain surfaces, which suggests that this chemical process is their most likely formation route. In addition, these models provide valuable constraints on the possible chemical pathways leading to the formation of complex organic molecules. Detecting the next member in the series of unbranched alkyl cyanides will indeed be a step forward in our understanding of the degree of chemical complexity that can be reached in the interstellar medium.
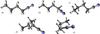 |
Fig. 1 The conformers of n-butyl cyanide. Those investigated in the present work are given in the upper row: a) anti-anti, b) gauche-anti, and c) anti-gauche; the others are in the lower row: d) gauche-gauche’ and e) gauche-gauche. The ordering is from a) being lowest to e) being highest in energy. The C and N atoms are indicated by gray and violet “spheres”, respectively, and the H atoms by small, light gray “spheres”. |
A complication of especially high-resolution spectroscopic investigations of n-butyl cyanide arises from the different conformations in which the molecule may occur. The anti-anti (or AA) conformer possesses a planar zig-zag structure of the heavy atoms as demonstrated in Fig. 1. Rotating either the cyano group or the terminal methyl group by about 120o yields the gauche-anti (GA) and anti-gauche (AG) conformer, respectively. Since either rotation can be performed clockwise as well as counterclockwise, both conformers are doubly degenerate with respect to the AA conformer. Rotating both groups yields two different conformers because they can be rotated to either the same or the opposite side; the former is called gauche-gauche (GG), while the latter is called gauche-gauche’ (GG’). Both conformers are also doubly degenerate with respect to the AA conformer.
Crowder (1989) studied the infrared spectrum of n-butyl cyanide in the gas phase. He stated that there are four spectroscopically distinguishable conformers. Among these, GA was found to be the most abundant one (46%), AA was slightly less abundant (30%), and even AG (13%) and a fourth one, presumably rather GG’ than GG (11%), were present. Molecular mechanics2 calculations yielded AA as being the lowest in energy with GA, AG, and GG’ being higher by 0.54, 3.68, and 4.31 kJ/mol (or 65, 443, and 519 K), respectively, roughly in accordance with the thermal population of the conformers. Very similar results were obtained for the isoelectronic 1-hexyne.
Bohn et al. (1997) studied the rotational spectrum of n-butyl cyanide between 5 GHz and 22 GHz employing molecular beam Fourier transform microwave (MB-FTMW) spectroscopy. Three conformers were identified, AA, GA, and AG. The first two were estimated to be lower in energy than the latter because of the intensities. These findings are in accordance with those from gas-phase infrared spectroscopy mentioned in the previous paragraph. More than 20 rotational transitions were recorded for each conformer with J and Ka extending to 8 and 2, respectively. 14N quadrupole splitting was observed for some transitions, and the diagonal components of the quadrupole tensor were determined.
Quantum chemical investigations into the energetics of the n-butyl cyanide conformers have been performed to our knowledge only by Atticks et al. (2002). They carried out Hartree-Fock (HF) and second order Møller-Plesset (MP2) ab-initio calculations for all five conformers of n-butyl cyanide. The HF calculations yielded AA and GA as low-energy conformers with AG and GG’ higher by about 4 kJ/mol (~500 K) and GG much higher by more than 8 kJ/mol (~ 1000 K). This energy ordering is compatible with both spectroscopic studies and is adopted in the present investigation. The higher level MP2 calculations lowered the relative energy of all conformers with a gauche orientation of the CN group, but raised the relative energy of AA. As a consequence, AA and GG’ were calculated to be essentially isoenergetic. These energies, however, are incompatible with experimental results and are thus discarded.
The previously obtained spectroscopic parameters for three conformers of n-butyl cyanide (Bohn et al. 1997) permit reliable predictions up to 40 GHz, partially even 60 GHz. However, already in the 3-mm region, deviations of a few megahertz must be expected. Therefore, we decided to investigate the rotational spectrum of this alkyl cyanide at shorter millimeter wavelengths to facilitate radio-astronomical observations throughout this region. The list of experimental transition frequencies was very greatly increased in the course of the present investigation, as were the frequency and quantum number ranges, permitting strong and medium strong transitions to be predicted accurately into the lower submillimeter-wave region. In addition, we used the data to search for the two lowest energy conformers of n-butyl cyanide in our 3 mm line survey of Sgr B2(N).
2. Experimental details
The spectral ranges 75−131 GHz and 200−223 GHz were studied with a new, fully solid-state, source-modulated spectrometer. Detection of the intrinsically weak rotational transitions exhibited by complex molecules required the integration of an absorption cell with an effective path length of 44 m. This double-pass cell comprises three Pyrex tubes of 10 cm diameter and almost equal lengths, and employs both a rooftop mirror, which rotates the polarization by 90 degrees, and a polarizing grid to separate the orthogonal source and detector beams. Millimeter-wave spectrometers with similar double-pass optics have been previously described in the literature, e. g. by Oh & Cohen (1992).
Observations of butyl cyanide absorption features were accomplished by establishing a slow gas flow through the absorption cell. This practice allowed the maintenance of the low and constant pressure (around 6 × 10-3 mbar) required to avoid pressure-broadening over long data acquisition times. Heating of the gas inlet valve to about 60 °C was also found to be necessary to avoid sample condensation.
The spectrometer’s radiation source consists of a microwave synthesizer covering the 10 to 43.5 GHz range whose output has been amplified, followed by a 3-mm band doubler and a chain of three 2-mm band doublers, respectively. The detector is a waveguide-mounted GaAs Schottky diode, optimally biased with a custom current source constructed at the Universität zu Köln. The detector output is processed by a lock-in amplifier with a time constant of 50 ms; digital averaging of six to eight points typically results in an effective time constant of 300−400 ms.
Additional measurements between 301 and 302 as well as between 342 GHz and 348 GHz employed a phase-locked backward-wave oscillator (BWO) spectrometer, the operating principles of which are described in greater detail in Winnewisser (1995) and Lewen et al. (1998). In the current spectrometer setup, phase locking is achieved by means of a harmonic mixer producing the third harmonic of a frequency-tripled microwave signal. The BWO beam thus stabilized in frequency is directed through a 3.4 m long, 10 cm diameter Pyrex cell and detected with a helium-cooled InSb hot-electron bolometer. Double-passing or larger cell dimensions are not needed here because of the high output power of the BWO (30 mW) and the low noise level of the bolometer. Frequency modulation and referencing are performed as described above.
3. Results
3.1. Laboratory spectroscopy
The rotational spectrum of a large molecule is denser than that of a smaller molecule
because the absorbed or emitted flux is distributed over more transitions spanning a
larger range of rotational quantum numbers J and
Ka, yielding larger partition function
values, and possibly over several conformers, increasing partition function values even
further. In addition, transitions are observed not only in the ground vibrational state,
but also in an increasing number of excited states for increasingly heavier molecules.
Some examples illustrate the increase in line density with increasing molecular
complexity. The rotational spectra of, e.g., CH3CN and SO2 are
comparatively sparse such that transitions of
13CH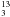 CN (Müller
et al. 2009) and SO17O (Müller et al.
2000), respectively, can be studied rather extensively by conventional absorption
spectroscopy in a sample of natural isotopic composition. And while 13C, and
even 15N, isotopologs of vinyl cyanide of natural isotopic composition were
studied (Müller et al. 2008; Kisiel et al. 2009), the 13C isotopologs of the only slightly
larger (2 H atoms) ethyl cyanide molecule were studied in isotopically enriched samples
(Demyk et al. 2007) because the increase in the
number of rotational states in each vibrational state as well as the increase in the
number of low-lying vibrational states result in a spectrum containing many more lines.
CN (Müller
et al. 2009) and SO17O (Müller et al.
2000), respectively, can be studied rather extensively by conventional absorption
spectroscopy in a sample of natural isotopic composition. And while 13C, and
even 15N, isotopologs of vinyl cyanide of natural isotopic composition were
studied (Müller et al. 2008; Kisiel et al. 2009), the 13C isotopologs of the only slightly
larger (2 H atoms) ethyl cyanide molecule were studied in isotopically enriched samples
(Demyk et al. 2007) because the increase in the
number of rotational states in each vibrational state as well as the increase in the
number of low-lying vibrational states result in a spectrum containing many more lines.
The rotational spectrum of n-butyl cyanide was studied in the microwave region initially by absorption spectroscopy by Bohn et al. (1997), but their spectrum turned out to be too dense to perform an in-depth analysis. These authors hence used MB-FTMW, which is a very good alternative way of studying the rotational spectrum of a large complex molecule, in particular for initial assignments, because low rotational temperatures, as low as 1 K, permit only a very small number of rotational levels to be populated compared to room temperature. Bohn et al. (1997) identified more than 20 transitions each between 5 GHz and 22 GHz for the AA, GA, and AG conformers with quantum numbers J and Ka up to 8 and 2, respectively. All three conformers are asymmetric tops where Ray’s asymmetry parameter κ = (2B − A − C)/(A − C) is close to the prolate limit of −1. The values for the AA, GA, and AG conformers are −0.9898, −0.9229, and −0.9866, respectively. The dipole moment components for each conformer were estimated from that of CH3CN by rotating the latter molecule such that the CN bonds are parallel. The authors obtained μa = 3.4 D and μb = 1.9 D for the AA conformer, and μc = 0 because of the symmetry. The values for the GA and AG conformers were μa = 2.3 D, μb = 3.1 D, and μc ≈ 0 D and μa = 3.7 D, μb ≈ 0 D, and μc = 1.2 D, respectively. The diagonal elements of the 14N quadrupole tensor were estimated equivalently from CH3CN data.
 |
Fig. 2 Section of the millimeter-wave spectrum of
n-C4H9CN displaying features caused by all
three conformers investigated in the present analysis. The
|
The rotational spectra of the AA, GA, and AG conformers of n-butyl cyanide were predicted and fit employing the SPCAT and SPFIT programs by Pickett (1991). The 14N hyperfine structure was not resolved in any of the present measurements because of the rather high quantum numbers and the larger line widths caused by using a free space cell rather than a molecular beam.
Our present measurements were started near 90 GHz. As shown in Fig. 2, the spectrum is very dense, and lines belonging to low-Ka transitions were displaced from the initial predictions by a few megahertz. The large number of lines recorded, the high line density, the presence of lines from three identified conformers, and very many unidentified lines made the analysis time-consuming and delicate particularly in the initial stages before improved predictions were available. Nevertheless, groups of closely spaced lines could be identified unambiguously because the predicted pattern was quite close to the observed one in terms of both frequencies and relative intensities. Improved predictions permitted us to assign transitions with higher Ka even if they were isolated. Only transitions were used in the fit for which overlap by unassignable lines or lines from other conformers appeared to be negligible. In the 75.0−130.4 GHz region, a-type R-branch transitions were used in the fit for all conformers. Many b-type transitions were included for the GA conformer, as can be expected from the large dipole moment component. In the case of the AA conformer, the smaller b-dipole moment component initially permitted only Ka = 1 ↔ 0 transitions to be assigned unambiguously in this frequency region. No c-type transitions were assignable unambiguously to the AG conformer throughout all frequency regions studied in the course of the present investigation as the dipole moment component is even smaller and the lines of this conformer were weaker because of the much lower abundance than those of the GA and AA conformers.
The J quantum number range was extended by additional measurements between 200.0 GHz and 222.3 GHz. It was straightforward to assign a-type transitions for all conformers as well as b-type transitions for the GA conformer. In addition, a fairly tight Ka = 8−7 Q-branch was predicted for the AA conformer in this frequency region whose relative intensity was quite favorable with respect to the lowest Kaa-type R-branch transitions. Several transitions of that Q-branch could be identified unambiguously and permitted other b-type transitions to be identified not only in this frequency region, but also in the 75.0−130.4 GHz region.
Finally, spectral recordings obtained near 300 GHz and 350 GHz were analyzed. Large sections of the Ka = 13−12 Q-branch of the AA conformer appeared to be overlapped negligibly by other features. In addition, some c-type transitions of the AG conformer appeared to be sufficiently close to the predictions and have about the right intensities, but their number are insufficiently large for us to make unambiguous assignments. Altogether, more than 2200 transitions each have been recorded in the present investigation for the AA and GA conformers, while almost 1400 transitions have been recorded for the higher energy AG conformer. The number of distinct frequencies is smaller by about a factor of two, mainly because of the unresolved asymmetry splitting. The highest J values of the transitions included in the fits are around 120 for the GA and AG conformers and even 136 for the AA conformer because of the smaller value of B + C. The highest values of Ka are higher than 40 for GA and AG, and reach a slightly lower value of 38 for the AA conformer because of the larger A rotational constant.
Among the previous MB-FTMW measurements, the 22,1−21,2 hyperfine components of the GA conformer were omitted from the fit because of the rather large and differing residuals. In addition, we excluded from the fit transitions with larger residuals for which no hyperfine splitting was resolved. These were the 808−707 transition of AA, the 707−606 transition of AG, and the 423−322 transition of GA.
Spectroscopic parameters were determined by employing Watson’s S reduction of the rotational Hamiltonian. The data from the previous MB-FTMW spectroscopic investigation (Bohn et al. 1997) were included in the fit with hyperfine splitting. Parameters were retained in the fit in general if they were determined with significance and if their inclusion contributed to the reduction in the rms error (the reduced χ2). The resulting spectroscopic parameters are given in Table 1. The transition frequencies with their assignments, uncertainties, and residuals between observed frequency and that calculated from the final set of spectroscopic parameters are available in the supplementary material, as outlined in the appendix, and are also available in the spectroscopy section3 of the CDMS (Müller et al. 2001, 2005).
The rms errors of the fits were slightly smaller than 1.0, meaning that the transition frequencies were reproduced on average within the experimental uncertainties. Moreover, partial rms errors for the MB-FTMW data and the present data around 100, 210, and above 300 GHz were mostly between 0.8 and 1.0 and did not exceed 1.1.
3.2. Radioastronomical observations
We used a complete line survey performed in the 3 mm atmospheric window between 80 and
116 GHz toward the hot core region Sgr B2(N). The observations were carried out in January
2004, September 2004, and January 2005 with the IRAM 30 m telescope on Pico Veleta, Spain.
Details about the observational setup and the data reduction are given in Belloche et al. (2008). An rms noise level of 15−20 mK
on the  scale was achieved below 100 GHz, 20−30 mK
between 100 GHz and 114.5 GHz, and about 50 mK between 114.5 GHz and 116 GHz.
scale was achieved below 100 GHz, 20−30 mK
between 100 GHz and 114.5 GHz, and about 50 mK between 114.5 GHz and 116 GHz.
This survey aims to investigate the molecular complexity of this prolific source and characterize its molecular content. Overall, we detected about 3700 lines above 3σ over the whole 3 mm band. These numbers correspond to an average line density of about 100 features per gigahertz. Given this high line density, the assignment of a line to a given molecule can be trusted only if all lines emitted by this molecule in our frequency coverage are detected with the correct intensity predicted by a model of its excitation and if none of the predicted lines are missing in the observed spectrum. It is possible to search for new species once a sufficient number of lines emitted by known molecules have been identified, including vibrationally and torsionally excited states. The XCLASS software4 is used to model the emission of all known molecules in the local thermodynamical equilibrium approximation (LTE for short), which refers here only to the rotational temperature from which the vibrational or conformational temperatures may differ. More details about this analysis are given in Belloche et al. (2008). About 50 different molecules have been identified in Sgr B2(N) thus far, and for several of them emission or absorption features due to minor isotopologs (about 60) or excited vibrational states (about 50) have also been identified (Belloche et al. in prep.). However, up to as many as 40% of the significant lines remain unassigned, and some of them are rather strong. The article reporting the results of the full survey will be submitted later this year (Belloche et al., in prep.), and the data will become public after acceptance of the article.
 |
Fig. 3 Sections of the 3 mm line survey of Sgr B2(N) showing the LTE modeling of the astronomical observations. In each panel, the black line represents the observed spectrum and the green line our current model of all firmly assigned species, i. e. without n-C4H9CN. The red line shows the model corresponding to the parameters listed in Table 2 for the AA conformer of n-butyl cyanide. |
Parameters of our best-fit LTE models of alkyl cyanides with upper limits for n-butyl cyanide, which was not unambiguously detected.
We searched for emission features of the AA and GA conformers of n-butyl cyanide in our molecular line survey because these are the two conformers lowest in energy. The AG conformer was deemed to be too high in energy to be observable under LTE conditions. Both AA and GA conformers were assumed to be ground state conformers since the exact energy difference between both conformers is unknown. As a consequence, the derived column density of the higher state conformer, GA according to present knowledge, or the upper limit thereof, is likely overestimated. The source size, rotational temperature, line width, and finally the velocity offset from the systemic velocity were assumed to be the same as those for the stronger hot-core component of n-propyl cyanide (see Table 2). The parameters of the lighter alkyl cyanides are only slightly different.
The partition function values at 300 K and 150 K for the vibrational ground states of the AA conformer of n-butyl cyanide were calculated as 174 758.2 and 61 661.6. The corresponding values for the GA conformer, 191 118.2 and 67 405.2, were calculated neglecting the conformational degeneracy as well as the non-zero energy of the lowest rotational state of the GA conformer. Vibrational contributions to the partition function were also neglected because no information is available on the low-lying vibrational states of n-butyl cyanide conformers. These contributions are probably not negligible at 150 K and may be very substantial at 300 K, where the latter is the default temperature for the catalog entries and corresponds roughly to the laboratory conditions, and the former is the probable temperature of n-butyl cyanide in Sgr B2(N) (see also Table 2). Additional partition function values will be provided in the CDMS.
Most transitions of both conformers are blended with stronger lines of other known species. However, a few transitions are relatively free of contamination and could in principle permit a detection. These are displayed in Figs. 3 and 4. Two transitions of the AA conformer coincide with lines that remain unidentified (Figs. 3b and c), but the prediction for another transition may be inconsistent with the observed spectrum depending on the exact position of the baseline, which is uncertain (Fig. 3a). As far as the GA conformer is concerned, two transitions coincide with still unidentified lines (Figs. 4a and c), one is partly blended with the weaker velocity component of a vibrationally excited state of C2H5CN (Fig. 4b), and one with vibrationally excited dimethyl ether (Fig. 4d).
Overall, the number of potentially detected emission lines of either n-butyl cyanide conformer is too small to warrant even a tentative detection. As a result, we consider the models displayed in Figs. 3 and 4 as upper limits to the emission of n-butyl cyanide. Therefore, we derive upper limits of 3.0 × 1015 cm-2 and 8.0 × 1015 cm-2 for the column densities of the AA and GA conformers of n-butyl cyanide, respectively (see Table 2).
4. Discussion
4.1. Laboratory spectroscopy
The present investigations permitted a full set of quartic centrifugal distortion parameters to be determined for the first time for each of the three n-butyl cyanide conformers previously characterized by MB-FTMW spectroscopy. In addition, several sextic distortion terms, up to a complete set for GA, along with some parameters of even higher order have been obtained with significance. Accurate predictions of the ground-state rotational spectrum are possible for strong to moderately weak transitions throughout the millimeter wave region and well into the submillimeter region for the two lower-energy conformers AA and GA. In the case of the higher-energy AG conformer, which was less well-characterized, predictions of a-type transitions should, nevertheless, be reliable up to possibly 500 GHz, but the c-type transitions are about as strong or stronger than the a-types beyond 300 GHz. However, they are difficult to assign because of the fairly large uncertainties for transitions with Ka higher than 5, and they are still weaker than the transitions of the two more abundant conformers.
Since the two conformers, for which no rotational transitions have been identified thus far, are even higher in energy, it may be difficult to identify them in the recorded spectra. The best prospects should be at lower frequencies because of the fewer as well as narrower lines.
The increase in DK and the decrease in both DJ and HJ from GA to AG and then AA reflect the increase in A and the decrease in B + C. A clear trend is also present for the HK values of the AA and GA conformers, permitting a value of 2.0 Hz to be estimated for the AG conformer. The value may be lower by at least 0.5 Hz or higher by at least 1.0 Hz. No clear trend is discernible for DJK as well as the higher-order diagonal distortion terms. Since GA is much farther away from the prolate limit than both AA and AG, off-diagonal distortion parameters, such as d1, d2, etc., play a much bigger role for this conformer than for the other two.
While almost all of the spectroscopic parameters of the three conformers have been firmly determined with small relative uncertainties, some parameters of the GA conformer were just barely determined, most notably l2 and PKKJ. However, the values of both parameters appear to be reasonable. The decline in magnitude from d1 to d2 is much more than a factor of 10, while it is about a factor of 10 from h1 to h2 and from h2 to h3. Hence, it is conceivable that l1 and l2 differ by a factor only slightly larger than 2. Similarly, there is a factor of around 10 000 from the magnitude of DJK to that of HKJ to LKKJ, and finally to that of PKKJ. It should be added that the correlation coefficients between l2 or PKKJ with any of the other spectroscopic parameters are rather small in magnitude, except for those between l2 and h2 and between PKKJ and LKKJ, which are fairly large, as expected. As a consequence, omission of l2 or PKKJ affected only the corresponding lower order parameters, in particular h2 or LKKJ, respectively. However, these changes were deemed to be too large with respect to their uncertainties, and l2 and PKKJ were retained in the fit. It is advisable to view their values with caution.
Infrared spectroscopy and low-level theoretical calculations (Crowder 1989; Atticks et al. 2002) predict the AA conformer to be the lowest in energy with the GA conformer being only slightly higher. The relative energies of the conformers could also be determined from relative intensity measurements in the rotational spectra. In addition to at least reasonable knowledge of the rotational and vibrational contributions to the partition function, one would need to know the dipole moment components accurately. In the case of monocyanides of alkanes or alkenes, the dipole vector is close to being aligned with the CN bond, as in the examples of either vinyl and ethyl cyanide (Kraśnicki & Kisiel 2011) or iso-propyl cyanide (Müller et al. 2011), and the total dipole moments are almost the same. The estimates of the dipole moment components for the n-butyl cyanide conformers, obtained by rotating the CH3CN dipole vector such that it is parallel to the CN bond of the respective conformer (Bohn et al. 1997), are hence probably quite good. Small deviations from the alignment with the CN bond, however, as well as uncertainties about the structure may have non-negligible effects on the values of the dipole moment components. Therefore, it is desirable to carry out Stark effect measurements on the n-butyl cyanide conformers, which we hope to be able to carry out soon.
We note that higher level (MP2) calculations overestimate the stability of the CN group in the gauche conformation compared to the anti conformation. Unsurprisingly, a similar situation was encountered for MP2 calculations on n-propyl cyanide by Traetteberg et al. (2000), who also describe a gas-phase electron-diffraction study of this molecule carried out at room temperature. The experimentally determined conformational composition, a gauche to anti ratio of about 3:1, was very similar to the one calculated theoretically. However, these results were recognized to be at variance with results from a microwave study (Wlodarczak et al. 1988) in which relative intensities were measured at room temperature and 233 K. The anti conformer was determined to be 1.1 ± 0.3 kJ/mol lower in energy than the gauche conformer. A detection of n-propyl cyanide in Sgr B2(N) by Belloche et al. (2009) was restricted to lines of the anti conformer, since transitions of the gauche conformer were too weak at a rotational temperature of 150 K to be detected unambiguously. It was concluded that the anti conformer may be even lower in energy than the gauche conformer or that the molecules are not in LTE.
The energetics of the n-butyl cyanide conformers suggest that a detection in space may only be possible for the AA conformer, that the GA conformer has some chance of being detected, but that the AG conformer, and even more so those not yet characterized by rotational spectroscopy, are likely too high in energy.
4.2. Radioastronomical observations
Since n-propyl cyanide has been detected thus far only in the Sgr B2(N) hot core, this source is presently the only viable source to search for n-butyl cyanide in space. Column density ratios of 108:80:1 were derived for methyl, ethyl, and n-propyl cyanide (Belloche et al. 2009). On the basis of the two heavier molecules, one would expect a column density drop of about two orders of magnitude for n-butyl cyanide with respect to n-propyl cyanide. However, since methyl and ethyl cyanide have almost equal column densities, the column density of n-butyl cyanide may also be quite similar to that of n-propyl cyanide. The actual determination of the column density of n-butyl cyanide in Sgr B2(N) will thus provide valuable clues to the formation of complex molecules in space.
The upper limits to the column density of the AA and GA conformers of n-butyl cyanide are lower by a factor of 5 and about 2, respectively, than the column density of n-propyl cyanide (see Table 2). The much lower, and thus more meaningful, upper limit to the AA conformer than the GA conformer can be explained by the smaller value of B + C of 2598 MHz versus 3343 MHz and the larger value of μa ≈ 3.4 D for AA versus μa ≈ 2.3 D and μb ≈ 3.1 D for GA (Bohn et al. 1997, see also Sect. 3.1). Since current knowledge indicates that the GA conformer is slightly higher in energy than the AA conformer, the column density of the GA conformer should be significantly less than twice that of the AA conformer at rotational temperatures significantly below room temperature5. If an energy difference of 0.54 kJ/mol (65 K) is assumed (Crowder 1989), which is compatible with the abundances derived from IR spectroscopy, the column density ratio between the AA and GA conformers should be about 1.0:1.2. On the basis of the upper limit of the AA conformer, an upper limit to the total n-butyl cyanide column density of 6.6 × 1015 cm-2 would be derived for the main component of Sgr B2(N). This is more than a factor of two lower than n-propyl cyanide. Hence, the possibility of very similar column densities of n-propyl cyanide and n-butyl cyanide, as mentioned above, may be unlikely. Decreasing the line confusion through interferometric observations should permit us to lower the column-density upper limit of n-butyl cyanide or could even lead to its detection. We hope to have observational results from ALMA soon.
Assigned transitions for the AA conformer of n-butyl cyanide, observed transition frequency (MHz), experimental uncertainty Unc. (MHz), residual O−C between observed frequency and that calculated from the final set of spectroscopic parameters, and weight for blended lines.
Concerning radioastronomical searches for n-butyl cyanide more generally, the molecules aminoacetonitrile (Belloche et al. 2008), n-propyl cyanide, and ethyl formate (Belloche et al. 2009) were detected most clearly at 3 mm because overlap with frequently stronger lines of lighter or more abundant molecules was not as significant in this frequency region as at either 2 or 1.3 mm. The frequencies are lower than or just reach the Boltzmann peaks of the a-type transitions at the derived rotational temperatures of 100 K for aminoacetonitrile (~ 230 GHz) and ethyl formate (~ 190 GHz) or 150 K for n-propyl cyanide (~ 200 GHz). With the Boltzmann peak for the a-type transitions of the AA conformer of n-butyl cyanide being at ~ 150 GHz at 150 K, the molecule is probably most reliably searched for with a single-dish telecope at wavelengths longer than 3 mm. Interferometric observations, e. g. with the Plateau de Bure Interferometer (PdBI, to be upgraded to NOEMA), the Expanded Very Large Array (EVLA), or the Atacama Large Millimeter Array (ALMA), will alleviate the line confusion problem somewhat, which may make searches for the AA conformer in space promising at 3 mm or maybe even shorter wavelengths. At any rate, the current laboratory measurements together with the previous ones as well as interpolations should cover all frequencies suitable for the search for n-butyl cyanide in space; moreover, extrapolation to higher frequencies is reasonable to some extent.
5. Conclusion
The rotational spectra of three low-lying conformers of n-butyl cyanide have been studied extensively in the millimeter and lower submillimeter regions, providing the means to search for this molecule in space. Inspection of our sensitive 3 mm line survey toward Sgr B2(N), carried out with the IRAM 30 m telescope, yielded an upper limit to the column density of the lowest energy AA conformer that is considerably lower than the column density found recently for the shorter n-propyl cyanide (Belloche et al. 2009). Observations with ALMA, other telescope arrays, or single-dish telescopes at lower frequencies should alleviate the line confusion, leading to a considerable lowering of the upper limit to or the actual detection of the molecule, either of which will provide important clues about the molecular complexity in space. Observational constraints on the column density of iso-propyl cyanide, a molecule that has also been studied recently in our laboratory (Müller et al. 2011), will also be interesting as it is the smallest branched cyanide. The detection of this molecule or sufficiently low upper limits to its column densities will provide important information on the importance of branched molecules with respect to their unbranched isomers. Several other complex molecules may also be detectable in this prolific source. It is thus likely that observations of Sgr B2(N) with ALMA around 3 mm will provide deeper insight into astrochemistry.
6. Appendix A. Supplementary material
The experimental transition frequencies for the AA, GA, and AG conformers of n-butyl cyanide are given as Tables 3, 4 and 5, respectively, in the supplementary material. Only the first and the last 10 lines of Table 3 are shown here; for complete versions of all conformers see the online tables at the CDS. The tables give the rotational quantum numbers J, Ka, and Kc for the upper state followed by those for the lower state. The 14N hyperfine structure was resolved at least in part in the previous measurements (Bohn et al. 1997), so the total spin-rotation quantum number F is given in addition in these cases for the upper and lower states. The observed transition frequency is given in megahertz units with its uncertainty and the residual between observed frequency and that calculated from the final set of spectroscopic parameters. The previous data extend to 22 GHz, and the data from the present investigation start at 75 GHz. Blended transitions are treated by fitting the intensity-averaged frequency, and this weight is also given in the tables. In most cases, the blending is caused by unresolved asymmetry splitting, i.e., the blended transitions agree in terms of their quantum numbers except for Kc (prolate paired transitions) or Ka (oblate paired transitions), and both transitions are equal in intensity. Accidental blending of transitions occured occasionally.
See e. g. the Molecules in Space page, http://www.astro.uni-koeln.de/cdms/molecules, of the Cologne Database for Molecular Spectroscopy, CDMS.
All n-butyl cyanide conformers other than AA are conformationally doubly degenerate, whereas AA is not degenerate (see Sect. 1).
Acknowledgments
We thank Prof. R. K. Bohn for providing the n-butyl cyanide transition frequencies with observed 14N quadrupole splitting. The present investigations have been supported by the Deutsche Forschungsgemeinschaft (DFG) in the framework of the collaborative research grant SFB 956, project B3. H.S.P.M. is very grateful to the Bundesministerium für Bildung und Forschung (BMBF) for financial support through project FKZ 50OF0901 (ICC HIFI Herschel) aimed at maintaining the Cologne Database for Molecular Spectroscopy, CDMS. This support has been administered by the Deutsches Zentrum für Luft- und Raumfahrt (DLR). A.W. and M.N. thank the French National Program PCMI (CNRS/INSU) for funding.
References
- Atticks, K. A., Bohn, R. K., & Michels, H. H. 2002, Int. J. Quant. Chem., 90, 1440 [CrossRef] [Google Scholar]
- Belloche, A., Menten, K. M., Comito, C., et al. 2008, A&A, 482, 179 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Belloche, A., Garrod, R. T., Müller, H. S. P., et al. 2009, A&A, 499, 215 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Bohn, R. K., Pardus, J. L., August, J., et al. 1997, J. Mol. Struct., 413, 293 [NASA ADS] [CrossRef] [Google Scholar]
- Crowder, G. A. 1989, J. Mol. Struct. (Theochem), 200, 235 [CrossRef] [Google Scholar]
- Demyk, K., Mäder, H., Tercero, B., et al. 2007, A&A, 466, 255 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Kisiel, Z., Pszczółkowski, L., Drouin, B. J., et al. 2009, J. Mol. Spectrosc., 258, 26 [NASA ADS] [CrossRef] [Google Scholar]
- Kraśnicki, A., & Kisiel, Z. 2011, J. Mol. Spectrosc., 270, 83 [Google Scholar]
- Lewen, F., Gendriesch, R., Pak, I., et al. 1998, Rev. Sci. Instrum., 69, 32 [NASA ADS] [CrossRef] [Google Scholar]
- Menten, K. M. 2004, in The Dense Interstellar Medium in Galaxies, Proc. 4th Cologne-Bonn-Zermatt Symp., Springer Proc. Physics, 91, 69 [Google Scholar]
- Müller, H. S. P., Farhoomand, J., Cohen, E. A., et al. 2000, J. Mol. Spectrosc., 201, 1 [NASA ADS] [CrossRef] [PubMed] [Google Scholar]
- Müller, H. S. P., Thorwirth, S., Roth, D. A., & Winnewisser, G. 2001, A&A, 370, L49 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Müller, H. S. P., Schlöder, F., Stutzki, J., & Winnewisser, G. 2005, J. Mol. Struct, 742, 215 [CrossRef] [Google Scholar]
- Müller, H. S. P., Belloche, A., Menten, K. M., et al. 2008, J. Mol. Spectrosc., 251, 319 [CrossRef] [Google Scholar]
- Müller, H. S. P., Drouin, B. J., & Pearson, J. C. 2009, A&A, 506, 1487 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Müller, H. S. P., Coutens, A., Walters, A., et al. 2011, J. Mol. Spectrosc., 267, 100 [CrossRef] [Google Scholar]
- Oh, J. J., & Cohen, E. A. 1992, J. Quant. Spec. Radiat. Transf., 48, 405 [NASA ADS] [CrossRef] [Google Scholar]
- Pickett, H. M. 1991, J. Mol. Spectrosc., 148, 371 [Google Scholar]
- Snyder, L. E., Kuan, Y.-J., & Miao, Y. 1994, LNP, 439, 187 [Google Scholar]
- Traetteberg, M., P. Bakken, P., & Hopf, H. 2000, J. Mol. Struct., 556, 189 [NASA ADS] [CrossRef] [Google Scholar]
- Winnewisser, G. 1995, Vib. Spectrosc., 8, 241 [CrossRef] [Google Scholar]
- Winnewisser, G., Krupnov, A. F., Tretyakov, M. Y., et al. 1994, J. Mol. Spectrosc., 165, 294 [NASA ADS] [CrossRef] [Google Scholar]
- Wlodarczak, G., Martinache, L., Demaison, J., et al. 1988, J. Mol. Spectrosc., 127, 178 [NASA ADS] [CrossRef] [Google Scholar]
All Tables
Parameters of our best-fit LTE models of alkyl cyanides with upper limits for n-butyl cyanide, which was not unambiguously detected.
Assigned transitions for the AA conformer of n-butyl cyanide, observed transition frequency (MHz), experimental uncertainty Unc. (MHz), residual O−C between observed frequency and that calculated from the final set of spectroscopic parameters, and weight for blended lines.
All Figures
 |
Fig. 1 The conformers of n-butyl cyanide. Those investigated in the present work are given in the upper row: a) anti-anti, b) gauche-anti, and c) anti-gauche; the others are in the lower row: d) gauche-gauche’ and e) gauche-gauche. The ordering is from a) being lowest to e) being highest in energy. The C and N atoms are indicated by gray and violet “spheres”, respectively, and the H atoms by small, light gray “spheres”. |
| In the text | |
 |
Fig. 2 Section of the millimeter-wave spectrum of
n-C4H9CN displaying features caused by all
three conformers investigated in the present analysis. The
|
| In the text | |
 |
Fig. 3 Sections of the 3 mm line survey of Sgr B2(N) showing the LTE modeling of the astronomical observations. In each panel, the black line represents the observed spectrum and the green line our current model of all firmly assigned species, i. e. without n-C4H9CN. The red line shows the model corresponding to the parameters listed in Table 2 for the AA conformer of n-butyl cyanide. |
| In the text | |
 |
Fig. 4 Same as Fig. 3 but for the slightly higher lying GA conformer of n-butyl cyanide. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.