| Issue |
A&A
Volume 462, Number 2, February I 2007
|
|
|---|---|---|
| Page(s) | 627 - 635 | |
| Section | Interstellar and circumstellar matter | |
| DOI | https://doi.org/10.1051/0004-6361:20066053 | |
| Published online | 02 November 2006 | |
Theoretical evaluation of PAH dication properties*
1
Centre d'Étude Spatiale des Rayonnements, CNRS et Université Paul Sabatier Toulouse 3, Observatoire Midi-Pyrénées, 9 avenue du Colonel Roche, 31028 Toulouse Cedex 04, France e-mail: [giuliano.malloci;christine.joblin]@cesr.fr
2
Istituto Nazionale di Astrofisica - Osservatorio Astronomico di Cagliari, Strada n.54, Loc. Poggio dei Pini, 09012 Capoterra (CA), Italy e-mail: [gmalloci;gmulas]@ca.astro.it
Received:
17
July
2006
Accepted:
25
September
2006
Aims.We present a systematic, theoretical study of 40 polycyclic aromatic hydrocarbon dications (PAHs++) containing up to 66 carbon atoms.
Methods.We performed our calculations using well-established quantum-chemical techniques in the framework of (i) the density functional theory (DFT) to obtain the electronic ground-state properties and of (ii) the time-dependent DFT (TD-DFT) to evaluate the excited-state properties.
Results.For each PAH++ considered, we computed the absolute visible-UV
photo-absorption cross-section up to about 30 eV. We also evaluated their
vibrational properties and compared them to those of the corresponding
neutral and singly-ionised species. We estimated the adiabatic and
vertical second ionisation energy 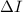 through total energy differences.
through total energy differences.
Conclusions.The 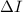 values obtained fall in the energy range 8-13 eV, confirming that
PAHs could reach the doubly-ionised state in HI regions.
The total integrated IR absorption cross-sections show a marked increase upon
ionisation, being on the average about two and five times larger for PAHs++
than for PAHs+ and PAHs, respectively.
The visible-UV photo-absorption cross-sections for the 0, +1 and +2
charge-states show comparable features but PAHs++ are found to absorb
slightly less than their parent neutral and singly ionised species between
~7 and ~12 eV. Combining these pieces of information, we found that
PAHs++ should actually be stabler against photodissociation than PAHs and
PAHs+, if dissociation thresholds are nearly unchanged by
ionisation.
values obtained fall in the energy range 8-13 eV, confirming that
PAHs could reach the doubly-ionised state in HI regions.
The total integrated IR absorption cross-sections show a marked increase upon
ionisation, being on the average about two and five times larger for PAHs++
than for PAHs+ and PAHs, respectively.
The visible-UV photo-absorption cross-sections for the 0, +1 and +2
charge-states show comparable features but PAHs++ are found to absorb
slightly less than their parent neutral and singly ionised species between
~7 and ~12 eV. Combining these pieces of information, we found that
PAHs++ should actually be stabler against photodissociation than PAHs and
PAHs+, if dissociation thresholds are nearly unchanged by
ionisation.
Key words: astrochemistry / molecular data / molecular processes / ISM: molecules / ultraviolet: ISM / infrared: ISM
© ESO, 2007
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


