Table A.1
Reaction energetic descriptors and comparison to the literature.
| Label | Reaction | ∆Urx | ∆U† | ∆U† Other Works | Tc (K) |
|---|---|---|---|---|---|
| H-addition reactions | |||||
| R1 | HCN + H• → H2CN• | -90.3 | 28.4 | 20.1a, 30.5b, 63.6c, 28.3d, 26.7f | 182 |
| R2-cis | HCN + H• → cis-HC•NH | -60.5 | 33.9 | 37.3a, 53.5b | 261 |
| R2-trans | HCN + H• → trans-HC•NH | -78.5 | 41.7 | 74.4c | 280 |
| R3 | HNC + H• → C•NH2 | -43.7 | 55.9 | 55.6a, 80.3c | 296 |
| R4-cis | HNC + H• → cis-HC•NH | -111.4 | 8.2 | 8.1a | 111 |
| R4-trans | HNC + H• → trans-HC•NH | -110.7 | 12.5 | 17.6c | 100 |
| R5 | H2CN• + H• → H2CNH | -406.4* | BL | BLa | |
| R6 | H2CN• + H• → 3[H3CN:] | -145.8 | 13.5 | 9.0a | 156 |
| R7 | C•NH2 + H• → 1[HC:NH2] | -383.9* | BL | BLa | |
| R8-cis | cis-HC•NH + H• → H2CNH | -450.8* | BL | BLa | |
| R8-trans | trans-HC•NH + H• → H2CNH | -445.1* | BL | ||
| R9-cis | cis-HC•NH + H• → 1[HC:NH2] | -328.7* | BL | BLa | |
| R9-trans | trans-HC•NH + H• → 1[HC:NH2] | -325.4* | BL | ||
| R10 | 3[H3CN:] + H• → H3CN•H | -385.1* | BL | BLa | |
| R11 | H2CNH + H• → H2CN•H2 | -145.9 | 22.9 | 19.4a, 25.5b, 12.7e, 17.7f | 186 |
| R12 | H2CNH + H• → H3CN•H | -128.8 | 16.0 | 15.1a, 19.2b | 171 |
| R13 | 1[HC:NH2] + H• → H2C•NH2 | -270.1 | 4.0 | N/Aa | 96 |
| R14 | 1[HC:NH2] + H• → HC•NH3 | -56.9 | 66.6 | 101.9 | 328 |
| R15 | H3CN•H + H• → CH3NH2 | -446.7* | BL | BLa | |
| R16 | H2C•NH2 + H• → CH3NH2 | -426.4* | BL | BLa | |
| H-abstraction and H2-addition reactions | |||||
| R17 | H2CN• + H• → HCN + H2 | -328.9 | BL | BLa | |
| R18 | C•NH2 + H• → HNC + H2 | -383.8* | BL | BLa | |
| R19-cis | cis-HC•NH + H• → HCN + H2 | -367.7 | BL | BLa | |
| R19-trans | trans-HC•NH + H• → HCN + H2 | -351.1 | 2.2 | 162 | |
| R20-cis | cis-HC•NH + H• → HNC + H2 | -335.8* | BL | BLa | |
| R20-trans | trans-HC•NH + H• → HNC + H2 | -320.7* | BL | ||
| R21 | 3[H3CN:] + H• → H2CN• + H2 | -279.6 | 6.7 | BLa | 135 |
| R22-cis | H2CNH + H• → cis-HC•NH + H2 | -33.9 | 24.8 | 45.0a | 340 |
| R22-trans | trans-HC•NH + H2 → H2CNH + H• | -24.7 | 36.6 | 371 | |
| R23 | H2CNH + H• → H2CN• + H2 | -54.0 | 29.4 | 406 | |
| R24-cis | 1[HC:NH2] + H• → cis-HC•NH + H2 | -138.5 | 28.3 | 22.7a | 349 |
| R24-trans | 1[HC:NH2] + H• → trans-HC•NH + H2 | -140.7 | 14.7 | 22.7a | 349 |
| R25 | 1[HC:NH2] + H• → C•NH2 + H2 | -68.1** | 8.8** | 3.5a | 177 |
| R26 | H3CN•H + H• → 3[H3CN:] + H2 | -73.0 | 15.6 | 12.9a | 356 |
| R27 | H3CN•H + H• → H2CNH + H2 | -301.7 | 4.8 | 1.8a | 129 |
| R28 | H2C•NH2 + H• → 1[HC:NH2] + H2 | -154.4* | BL | N/Aa | |
| R29 | H2C•NH2 + H• → H2CNH + H2 | -273.5 | BL | N/Aa | |
| R30 | HC•NH3 + H• → 1[HC:NH2] + H2 | -373.6 | BL | ||
| R31 | HC•NH3 + H• → 3[:CNH3] + H2 | -52.7** | 6.6** | 57 | |
| R32 | CH3NH2 + H• → H2C•NH2 + H2 | -67.3, -38.1 | 9.9, 30.2 | 37.3a, 33.4e | 125, 337 |
| R33 | CH3NH2 + H• → H3CN•H + H2 | -14.3 | 48.0 | 44.8a, 39.7e | 414 |
| R34 | 1[HC:NH2] + H2 → CH3NH2† | -231.7 | 102.6 | 270 | |
| Water-assisted H-transfer and isomerization reactions | |||||
| R35 | HNC 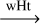 HCN HCN |
-32.4, -33.7 | 35.1, 73.6 | 49.6h | 221, 149 |
| R36 | trans-HC•NH 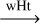 H2CN• H2CN•
|
-32.9 | 83.7 | ||
| R37-isom | cis-HC•NH 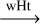 trans-HC•NH trans-HC•NH |
-9.6 | 23.9 | 136 | |
| R37-wHt | cis-HC•NH 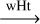 trans-HC•NH trans-HC•NH |
-22.6 | 65.5 | 196 | |
| R38 | C•NH2 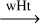 trans-HC•NH trans-HC•NH |
-47.1 | 30.8 | 88.8a | 52 |
| R39 |
1[HC:NH2] 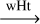 H2CNH H2CNH |
-106.1 | 4.9 | 22.7a, 14.0g | 187 |
| R40 | H3CN•H 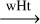 H2C•NH2 H2C•NH2
|
-21.5 | 69.3 | 100 | |
| R41 | HC•NH3 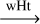 H2C•NH2 H2C•NH2
|
-209.3 | BL | BLa | |
a Molpeceres et al. (2024) (ice), b Woon (2002) (gas), c Talbi & Ellinger (1996), d Majumdar et al. (2018) (gas), e Joshi & Lee (2022) (gas), f de Jesus et al. (2021) (gas), g Ferrero et al. (2024) (ice), h Baiano et al. (2022).
*Calculated with respect to the asymptote, omitting ZPE corrections.
**Calculated with ωB97m-D3(BJ)/ma-def2-TZVP due to unrealistic ZPE correction at M062X-D3(0).
†The reaction does not stop at H2CNH2 + H, but it directly evolves to methylamine over a very high barrier.
Reaction R32 appears twice in the table because two different binding configurations of the reactants were identified, leading to distinct activation barriers (see Sect. 4).
Reaction R35 is also listed twice, corresponding to water-assisted H-transfer (wHt) mechanisms involving three or two water molecules, respectively.
The reactions 3[:CNH3] + H• → HC•NH3 and HC•NH3  H2C•NH2 (R42 and R43) are not included in this table, as they ultimately lead to H2C•NH2 after direct optimization of HC•NH3 in our model.
H2C•NH2 (R42 and R43) are not included in this table, as they ultimately lead to H2C•NH2 after direct optimization of HC•NH3 in our model.
Notes. Summary of the reactions considered in this work and their energetics. Each reaction is assigned a label (R1, R2,...) that is used throughout the text. Energy units are in kJ mol−1. ∆Urx and ∆U‡ are the reaction and activation energies of each reaction, Tc is the tunneling crossover temperature. BL stands for barrier-less and N/A for “not an answer.”
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


