| Issue |
A&A
Volume 708, April 2026
|
|
|---|---|---|
| Article Number | A281 | |
| Number of page(s) | 10 | |
| Section | Interstellar and circumstellar matter | |
| DOI | https://doi.org/10.1051/0004-6361/202658985 | |
| Published online | 16 April 2026 | |
C5H6 formation in Dense Molecular Clouds
1
Université Paris-Saclay, CNRS, Institut des Sciences Moléculaires d’Orsay,
91405
Orsay,
France
2
Université Bordeaux, CNRS, Bordeaux INP, ISM, UMR 5255,
33400
Talence,
France
3
Department of Physics and Astronomy, Aarhus University,
Ny Munkegade 120,
8000
Aarhus C,
Denmark
4
Université Paris-Saclay, CNRS, Institut de Chimie Physique, UMR8000,
91405
Orsay,
France
5
Synchrotron SOLEIL, L’Orme des Merisiers,
91192
Saint Aubin, Gif-sur-Yvette,
France
6
Department of Physics, University of Trento,
Via Sommarive 14,
38123
Trento,
Italy
★ Corresponding authors: This email address is being protected from spambots. You need JavaScript enabled to view it.
; This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
15
January
2026
Accepted:
16
February
2026
Abstract
Context. Cyclopentadiene (C5H6) has been recognized as a crucial precursor in the formation of nonplanar polycyclic aromatic hydrocarbons (PAHs) and carbon-rich nanostructures in space. Despite its significance and detection in the Taurus Molecular Cloud (TMC-1), the elementary gas-phase reaction pathways leading to cyclopentadiene from acyclic hydrocarbon precursors remain poorly constrained. This gap is emphasized by persistent discrepancies between astrochemical model predictions and the abundances inferred from its radioastronomical detection.
Aims. The aim of this work is to reconcile the chemical network that predicts the formation of C5H6 – a key intermediate in the growth of aromatic species – from gas-phase chemistry with its observed abundance in TMC-1.
Methods. We used combined experimental work conducted with the CERISES tandem mass spectrometer at the SOLEIL synchrotron, together with quantum chemical calculations, to determine and refine reaction rate coefficients and branching ratios for reaction pathways that lead to the formation of C5H7+. We then incorporated these results into the gas–grain chemical code NAUTILUS to model the formation of C5H6.
Results. We identify the reactions C2H4+ + CH3CCH and C3H7+ + C2H2 as an important source of C5H7+ and, consequently, of C5H6 in TMC-1. We experimentally determined the C2H4+ + CH3CCH reaction rate to be 1 × 10−9 cm3 s−1. In addition, the radiative association reactions C4H5 + H and C5H5 + H under low-pressure conditions deserve further investigation, as they may constitute key intermediate steps in the formation of C5H6 through neutral–neutral reaction pathways.
Conclusions. Our updated chemical model accounts for several previously missing formation pathways of C5H6. Although it reproduces only ~20% of the observed abundance, this represents a significant improvement compared to existing models. We identify the reactions C2H4+ + CH3CCH and C3H7+ + C2H2 as the two dominant sources of C5H7+. However, the formation of C5H6 through neu-tral-neutral chemistry remains poorly constrained. The main source is the radiative association of C5H5 + H but 1,3-butadiene (C4H6) appears to be a key intermediate in the formation of C5H6; however, its abundance is uncertain due to the disputed detection of its cyano-derivative proxy. Further work is therefore required to better constrain the abundance of 1,3-butadiene, which may be efficiently formed through the radiative association C4H5 + H.
Key words: astrochemistry / methods: laboratory: molecular / ISM: abundances / ISM: molecules / ISM: individual objects: TMC-1
© The Authors 2026
 Open Access article, published by EDP Sciences, under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Open Access article, published by EDP Sciences, under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article is published in open access under the Subscribe to Open model. This email address is being protected from spambots. You need JavaScript enabled to view it. to support open access publication.
1 Introduction
Cold molecular clouds, exemplified by the Taurus Molecular Cloud (TMC-1), have long been regarded as prime astrophysical environments for probing molecular mass growth on macroscopic scales. Through the combination of high-sensitivity astronomical observations and astrochemical modeling based on extensive gas-phase reaction networks involving ion–molecule and neutral–neutral processes, these regions provide unique insights into the formation pathways of complex molecules.
Following the detection of cyclic C3H2 by Thaddeus et al. (1985), decades of sustained effort have been devoted to the identification of aromatic and cyclic hydrocarbons in the interstellar medium (ISM), with particularly significant success achieved only recently in the cold dark cloud TMC-1. This advancement has been driven by large observational programs—most notably the QUIJOTE and GOTHAM surveys—which have played a key role in substantially expanding the known inventory of cyclic hydrocarbons in space. These efforts have resulted in the detection of a wide variety of nonaromatic and aromatic species, including ethynyl cyclopropenylidene (C3HCCH), cyclopentadiene (C5H6), and indene (C9H8) in Cernicharo et al. (2021a), ethynylbenzene (C6H5CCH) by Loru et al. (2023), o-benzyne (o-C6H4) by Cernicharo et al. (2021b), fulvenallene (C5H4CCH2) by Cernicharo et al. (2022), and phenalene (C13H10) by Cabezas et al. (2025).
In addition, numerous cyano derivatives have been identified, including species whose hydrocarbon counterparts are undetectable, as well as derivatives of already detected cyclic molecules. Notable examples include benzonitrile (C6H5CN) by McGuire et al. (2018), cyanocyclopentadiene (C5H5CN) by McCarthy et al. (2021), cyanonaphthalene (C10H7CN) by McGuire et al. (2021), cyanoindene (C9H7CN) by Sita et al. (2022), cyanoacenaphthylene (C12H7CN) by Cernicharo et al. (2024), cyanopyrene (C16H9CN) by Wenzel et al. (2024), and cyanocoronene (C24H11CN) by Wenzel et al. (2025). Many of these molecules exhibit relatively high fractional abundances, reaching up to 10−8 with respect to molecular hydrogen, which has fueled growing interest in the elementary chemical mechanisms that drive the conversion of acyclic hydrocarbons into cyclic species and, ultimately, polycyclic aromatic hydrocarbons (PAHs).
Within this molecular inventory, C5H6 occupies a pivotal role. It serves as a key precursor to five-membered-ringcontaining aromatic molecules such as indene (C9H8), which themselves are fundamental structural units in the formation of bowl-shaped aromatic hydrocarbons and carbon cages. Consequently, any comprehensive description of molecular mass growth toward nonplanar aromatic systems must begin with a detailed understanding of how cyclopentadiene is synthesized from simpler, acyclic precursors.
Despite its detection in TMC-1, robust gas-phase formation mechanisms for cyclopentadiene remain poorly constrained. As a result, current classical ion-molecule-based astrochemical models significantly underpredict its abundance, by several orders of magnitude, relative to astronomical observations (Cernicharo et al. 2021a; Mallo et al. 2025). This discrepancy highlights a critical gap in our knowledge of synthetic pathways to cyclopentadiene under interstellar conditions. Identifying and characterizing the missing pathways is therefore essential for advancing our understanding of molecular mass growth in cold clouds and for elucidating the chemical evolution leading to complex PAHs that could ultimately lead to carbonaceous dust grains.
Two distinct formation pathways have been proposed for C5H6. The most recent involves a nonclassical neutral–neutral chemistry developed by Yang et al. (2024). The more conventional pathway proceeds through a sequence of ion–molecule reactions that lead to the protonated precursor C5H7+, which subsequently forms C5H6 via dissociative recombination (DR), in analogy with the formation of benzene from C6H7+(Loison et al. 2026). However, astrochemical models relying on this latter pathway systematically underestimate the observed abundance of C5H6, which likely indicates the absence of one or more key ion–molecule reactions that efficiently produce C5H7+, as noted by Mallo et al. (2025).
In contrast, the model based on neutral–neutral chemistry proposed by Yang et al. (2024) appears to approach the observed abundance of C5H6. In this model, the dominant pathway is the reaction CH + C4H6 → C5H6 + H, where C4H6 corresponds to 1,3-butadiene. A significant limitation of this pathway arises from the uncertainty in the abundance of C4H6. While the abundance of the CH radical is well constrained by observations, C4H6 remains undetected due to its lack of a permanent dipole moment, and its abundance is inferred solely through chemical modeling (Cooke et al. 2023; Agúndez et al. 2025). Moreover, the model of Yang et al. (2024) neglects the reaction of C4H6 with atomic carbon, which is expected to represent a major destruction route for C4H6 (Husain & Ioannou 1997; Hahndorf et al. 2000), and then the model likely overestimates the C4H6 abundance.
In this study, we therefore conducted another exploration of potential ion–molecule reactions leading to C5H7+. The most promising candidate reaction was subsequently investigated experimentally, and the resulting new kinetic data were incorporated into the astrochemical model.
2 Methods
2.1 Experimental
We produced parent ions (C2H4+) via direct photoionization of ethylene (N35 air liquide) using vacuum ultraviolet (VUV) radiation in the 10.5–12 eV range at the DESIRS beamline of the SOLEIL synchrotron (Saint-Aubin, France). We investigated the reactions using the CERISES apparatus, which is a home-built tandem mass spectrometer coupled to the VUV source (Cunha De Miranda et al. 2015; Rossi et al. 2023). Following mass selection, the parent ions were guided into a reaction cell with a controlled collision energy, and the resulting product ions were analyzed using a second quadrupole. We derived absolute reaction cross sections from the neutral reactant (propyne and allene) pressure and measured ion intensities. The pressure was kept as low as possible (below 100 nbar) to maximize the likelihood of single-collision conditions. We examined the reaction with both C3H4 isomers at a fixed center-of-mass collision energy of 0.15 eV, while the internal energy of the parent ions was varied by tuning the photon energy. We determined the rate constants from the absolute reaction cross section measured, following the methodology described in the study of C3H+ reactivity by Rossi et al. (2025).
2.2 Astrochemical modeling
We employed a standard astrochemical model using the Nautilus code (Ruaud et al. 2016), which is a three-phase, time-dependent, chemical model that includes the gas phase, the dust-grain ice surface, and the dust-grain ice mantle. The model is based on the kida.uva.2024 reaction network (Wakelam et al. 2024), which has been recently updated to provide an improved description of complex organic molecule (COM) chemistry both on interstellar dust grains and in the gas phase (Manigand et al. 2021; Hickson et al. 2021; Coutens et al. 2022; Hickson et al. 2024). There are 800 individual species included in the network that are involved in approximately 9000 separate reactions. Elements are initially assumed to be in their atomic or ionic forms, and elements that have an ionization potential lower than 13.6 eV are considered to be fully ionized. This assumption reflects the adopted initial abundances at time t = 0 in the evolution of dense clouds, which are taken to correspond to those of a diffuse cloud. In such environments, elements are predominantly in their atomic form, and ultraviolet photons ionize species with ionization potentials below 13.6 eV. The high abundance of atomic hydrogen strongly limits the propagation of photons with energies above 13.6 eV, as these photons are efficiently absorbed by hydrogen atoms. The C/O gas phase elemental ratio is equal to 1 in this work. This value, which is higher than the cosmic abundance ratio (0.6), leads to a significantly better agreement between the model predictions and the full set of observations. For lower C/O ratios, the available carbon is insufficient to bind most of the oxygen into CO, leaving excess oxygen atoms that suppress radical abundances and consequently inhibit the chemistry. This choice is consistent with that adopted in previous studies of aromatic species in dense molecular clouds, including Hincelin et al. (2011), Byrne et al. (2024) and Mallo et al. (2025). Physically, this oxygen depletion is attributed to the formation of water ice during the history of the dense cloud. This water ice remains largely trapped on dust grains and does not desorb during subsequent warmer and less dense phases, in contrast to CO, which efficiently desorbs and is dissociated into C and O under these conditions (Hincelin et al. 2016; Wakelam et al. 2019). The initial simulation parameters adopted in this work are summarized in Table A.1. The grain surface and the mantle are both chemically active for these simulations, while accretion and desorption are only allowed between the surface and the gas phase. The dust-to-gas ratio (in terms of mass) is 0.01. A sticking probability of 1 is assumed for all neutral species, while desorption occurs by both thermal and nonthermal processes (cosmic rays, chemical desorption), including sputtering of ices by cosmic ray collisions (Wakelam et al. 2021).
2.3 Identification of important reactions
We identified Potential formation routes of C5H6 following an approach similar to that adopted in previous studies of Titan’s atmospheric chemistry (Hébrard et al. 2012, 2013; Dobrijevic et al. 2014; Loison et al. 2015; Dobrijevic et al. 2016; Loison et al. 2019) and of carbon chemistry in dense molecular clouds (Loison et al. 2013, 2014, 2016, 2017). We focused on the analysis of reactions leading to species directly related to C5H6, namely C5H5, C5H6, and C5H7+, and restricted to pathways involving species detected or predicted to have significant modeled abundances. We considered both ion–molecule and neutral–neutral reactions. For ionic chemistry, we examined systems of the type CHx+ + C4Hy, C2Hx+ + C3Hy, and related reactions, together with neutral reactions such as CHx + C4Hy. Species with negligible abundances, notably CH+ and CH2+, which are rapidly removed by reactions with H2, were excluded as potential precursors of aromatic C5H6. Once a comprehensive list of reactions that potentially produce C5H5, C5H6, and C5H7+ had been compiled, we conducted an extensive bibliographic search for each reaction. When reaction rate coefficients had been measured, at least at room temperature, and were found to be larger than 10−11 cm3 s−1, these reactions were assumed to be barrierless and therefore efficient under dense molecular cloud conditions. For many reactions, no experimental or theoretical data were available. In such cases, reactions that could plausibly be fast at low temperatures were first identified and we systematically investigated the presence of activation barriers by searching for transition states (TS) using density functionnal theory (DFT) calculations performed using the Gaussian 16 software package (Frisch et al. 2016). We employed the M06-2X functional with the cc-pVTZ basis set. This highly nonlocal functional, developed by Zhao & Truhlar (2008), is well suited for describing transition-state structures and energetics. However, the absence of a located TS does not necessarily imply the absence of an activation barrier. Reactions for which no TS was found were therefore compared with analogous, well-characterized reactions. For example, we assumed that if a species reacts with C2H2, it is also likely to react with other alkynes, and similarly that reactivity with C2H4 implies reactivity with alkenes. For reactions considered barrierless, we determined or estimated reaction products and branching ratios. When experimental data were unavailable, we used existing theoretical studies when possible. In the absence of any prior information, we performed additional DFT calculations to characterize key intermediates, allowing likely products and branching ratios to be estimated. It should be noted that, given the number of atoms involved and the excess energy available in these systems, a very large number of intermediate species may be formed. The present approach does not aim to identify all possible intermediates, which would require detailed, reaction-specific studies, such as those performed for the l–C3H3+ + C2H4 reaction by Mallo et al. (2025). Instead, we assess the feasibility of cyclization and the formation of aromatic compounds.
 |
Fig. 1 Panels a and c display the C2H4+ parent-ion signal from the photoionization of ethylene. Panels b and d show the reaction cross sections (left axis) and rate constants (right axis) with propyne and allene, respectively, as a function of photon energy. |
3 Experimental investigation of the reaction C2H4+ + C3H4
Once the reaction of C2H4+ with propyne was identified as a potentially important chemical route to C5H7+, we decided to experimentally investigate its reaction rate and branching ratio as a function of internal energy. Although allene is not directly detected due to its lack of a permanent dipole moment, it is assumed to be as abundant as propyne in TMC-1. We therefore investigated the reaction of C2H4+ with both C3H4 isomers.
Panels b and d in Figure 1 present the reaction cross sections (left axis) and rate constants (right axis) for reactions with propyne and allene, respectively, as a function of photon energy. Only reaction channels with cross sections exceeding those leading to C6H7+ are shown (3 Å2 and 7 Å2 at 10.6 eV for reactions with propyne and allene, respectively). The C6H7+ signal arises from a secondary process, namely charge transfer followed by the reaction C3H4+ + C3H4. Since C3H4+ is formed via charge transfer with no transfer of momentum, it has a sufficiently long residence time in the reaction cell to undergo secondary reactions with C3H4, such that this channel cannot be fully suppressed even at very low pressure. However, all nonreported channels contribute, at maximum, a few percent relative to the dominant product, as can be seen in Fig. B.1. Overall, we observed no significant dependence on photon energy over the explored range. Consequently, the reaction rates reported in Appendix C were taken at the lowest photon energy (10.6 eV), which corresponds to the minimum possible internal energy of the ions.
Three products dominate the reaction with propyne (panel b): the formation of C5H7+ (m/z 67), the charge-transfer product C3H4+ (m/z 40), and C4H5+ (m/z 53). All reaction rates are listed in the table in Appendix C, row 6. In contrast, the reaction with allene predominantly yields the charge-transfer product, with a rate that is more than an order of magnitude higher than the formation of C5H7+. Corresponding data are provided in the table in Appendix C, row 7.
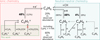 |
Fig. 2 Schematic diagram showing the main formation routes of C5H6 in TMC-1. Percentages written in bold indicate the contribution of the reactions in the nominal model. Percentages in normal text, associated linked with dashed arrows, correspond to their contribution if the C4H5 + H reaction is included. |
4 Formation of C5H6 in TMC-1
Figure 2 summarizes the main formation routes of C5H6 in TMC-1. Two comparable contributions are identified in the nominal model: the ion–molecule network and the neutral–neutral chemistry, each accounting for about 50% of the total production based on experimentally measured or theoretically validated rate coefficients. The ionic contribution is dominated by the reactions C2H4+ + CH3CCH and C3H7+ + C2H2, which are discussed in detail below. In the neutral network, the dominant source is the radiative association (RA) C5H5 + H. The alternative pathway C6H4 + CH, recently proposed by Yang et al. (2024), is reassessed here. The possible indirect role of the RA reaction C4H5 + H is also examined. The complete list of reactions considered is presented in the table in Appendix C.
4.1 Ionic chemistry
In the chemical network, ion–molecule chemistry contributes to the formation of C5H6 primarily through the production of the precursor ion C5H7+, followed by DR. Although no direct measurements of branching ratios for the DR of aromatic ions are currently available, laboratory studies of C6D6+ and C6D7+ show that the aromatic ring is preserved in more than 90% of events (Hamberg et al. 2011). By analogy, we assume that ring preservation also dominates the DR of C5H7+, such that the C5H7+ + e− reaction predominantly yields C5H6 + H. A comparison between model predictions and observations of C5H6 is shown in Fig. 3, with panel (a) illustrating the relative contribution of ionic chemistry to the overall formation of C5H6.
The C5H7+ ion is produced mainly through two reactions: C2H4+ + CH3CCH, which has been studied both here and previously (Anicich et al. 2006), and C3H7+ + C2H2. To date, no dedicated experimental or theoretical investigation of the latter reaction has been reported. However, it is isoelectronic with C3H5+ + C2H4 (Anicich et al. 2003, 2006; McEwan et al. 1998) and exhibits similar exothermicities, which suggests that it is a viable and potentially efficient source of C5H7+. Preliminary calculations performed in this work indicate that the CH3CH2CH2+ isomer reacts with C2H2 to form C5H7+ + H2, which supports this interpretation and highlights the need for more detailed studies. Several additional formation routes to C5H7+ were identified. Some are experimentally well characterized, including C2H5+ + CH3CCH/CH2CCH2 (Lifshitz et al. 1981), C3H4+ + C2H4 (McEwan et al. 1998; Anicich et al. 2006), and C3H5+ + C2H4 (Anicich et al. 2003, 2006; McEwan et al. 1998). Other plausible contributors include CH3+ + CH2CHCHCH2, C2H3+ + C3H6, C3H6+ + C2H2, and C4H3+ + C3H6, although these remain poorly constrained.
By contrast, the O + C6H7+ reaction does not represent a viable source of C5H7+. An activation barrier of approximately 7 kJ mol−1 was identified at the M06-2X/AVTZ level in this work. While this level of theory is generally reliable for locating transition states, it is known to underestimate barriers in reactions involving atomic oxygen, which reinforces the conclusion that this pathway is inefficient under dense cloud conditions.
Finally, although many reactions in the network produce C5H5+ ions, either as the most stable isomer c–C3H2CHCH2+ or as five-membered-ring structures, these species do not contribute to C5H6 formation. The various C5H5+ isomers react inefficiently with H2 (Mallo et al. (2025) and this work), and therefore cannot form C5H7+, which excludes them as viable precursors of cyclopentadiene.
 |
Fig. 3 Gas–grain astrochemical model results for C5H6 in dark clouds as a function of cloud age. (a–c) Nominal network results (solid black line); nominal network but with only some specific reactions as a source of C5H6 (solid red line). (d) Nominal with H + C4H5 leading to C4H6 (solid black line), nominal with H + C4H5 leading to C4H6 with only H + C5H5 leading to C5H6 (solid red line), and nominal with H + C4H5 leading to C4H6 with only CH + C4H6 leading to C5H6 (solid blue line). The horizontal gray region represents the observed abundances in TMC-1 with an arbitrary error associated (±2). The estimated age of the cloud, marked by a vertical gray region, corresponds to the time at which our model predictions best match the observed abundances of 67 key species. |
4.2 Neutral–neutral chemistry
In addition to ion–molecule processes, C5H6 can also be formed through neutral–neutral chemistry. Two such pathways contribute in the model, with the dominant route being the RA reaction H + C5H5 (48%), and a secondary contribution arising from CH + C4H6 (4%), as can be seen in Figure 3, panels b and c, respectively.
The H + C5H5 reaction plays a central role because the C5H5 radical is efficiently produced through several channels, most notably O + C6H5 (Frank et al. 1994), and potentially through H-assisted isomerization of CH2CHCHCCH. The latter species is itself efficiently formed via the C + C4H6 reaction (Husain & Ioannou 1997; Hahndorf et al. 2000). Rate coefficients for the H + C5H5 RA reaction can be estimated from previous experimental and theoretical studies (Frank et al. 1994; Harding et al. 2007; Vuitton et al. 2011). When this pathway is included, the modeled abundance of C5H6 is in relatively good agreement with observations, as shown in Figure 3b.
The secondary neutral pathway, CH + C4H6, has been investigated previously (He et al. 2020; McCarthy et al. 2021; Cernicharo et al. 2022). In our chemical network, the modeled abundance of CH closely matches observations (Suutarinen et al. 2011). However, C4H6 is mainly produced through CH + C3H6 (Daugey et al. 2005; Loison & Bergeat 2009; Zhou et al. 2025), with a minor contribution from C + C3H7. As a result, the modeled abundance of C4H6 remains low, in agreement with Cooke et al. (2023); Agúndez et al. (2025), making CH + C4H6 only a minor source of C5H6 as shown in Figure 3c) in contrast to the conclusions of Yang et al. (2024).
If, however, the reaction H + C4H5 were to produce C4H6 efficiently at the low pressures of dense molecular clouds, at rates approaching those calculated by Harding et al. (2007) for high-pressure conditions, the abundance of C4H6 would increase substantially. In that case, the CH + C4H6 reaction becomes more important, although it remains a secondary source of C5H6. The improved agreement with observations in this scenario primarily arises from the concomitant enhancement of the H + C5H5 RA pathway, as higher C4H6 abundances also lead to increased production of C5H5 isomers, as shown in Figure 3d. However, this scenario also leads to a modeled abundance of CH2CHCHCH2 of 1.6 × 10−9 at the chemical age of TMC-1, which exceeds the upper limit inferred from the non-detection of C4H5CN (Agúndez et al. 2025) by an order of magnitude. For this reason, our nominal model assumes that H + C4H5 proceeds exclusively toward CH3 + C3H3, following the ab initio calculations of Lee et al. (2003), despite the high-pressure results of Harding et al. (2007). The importance of the H + C4H5 reaction stems from the efficient formation of C4H5 radicals (in their various isomeric forms) via C + C3H6 (Chastaing et al. 1999; Loison & Bergeat 2004; Chin et al. 2013; Capron et al. 2015).
Finally, the prominence of hydrogen-atom reactions, including RA, is reinforced by the high and nearly constant abundance of atomic hydrogen in dense molecular clouds. Although H atoms readily stick to grains, they are continuously replenished by the cosmic-ray-induced dissociation of H2. In contrast, heavier atoms such as C, N, and O are efficiently depleted from the gas phase through grain sticking and, in the case of C and O, conversion into CO after ~105 years, further emphasizing the long-term importance of H-driven neutral chemistry.
5 Conclusions
In this work, we revisited the gas-phase chemistry that leads to the formation of cyclopentadiene (C5H6) in the cold dark cloud TMC-1 by adopting an integrated strategy that combines new laboratory measurements, quantum chemical calculations, astrochemical modeling, and chemical intuition to guide the identification of the most relevant reaction pathways. In particular, we experimentally characterized the reaction of C2H4+ with propyne and allene and incorporated the resulting reaction rate coefficients and branching ratios into an updated chemical network. This synergistic approach allowed us to identify previously underappreciated ion–molecule pathways that contribute to the formation of the key precursor ion C5H7+, and to reassess the relative roles of ionic and neutral–neutral chemistry in the synthesis of C5H6.
Our updated model partially alleviates the discrepancy between observed and modeled abundances of cyclopentadiene but still reproduces only about 20% of the abundance inferred from observations of TMC-1. Among the ionic routes, the reactions C2H4+ + CH3CCH and C3H7+ + C2H2 emerge as the dominant sources of C5H7+, highlighting the importance of relatively small hydrocarbon ions reacting with abundant unsaturated neutral species. However, even when these channels are treated with experimentally constrained rate coefficients, the overall efficiency of the ion–molecule network remains insufficient to fully account for the observed abundance of C5H6.
Neutral–neutral chemistry therefore appears to play a critical complementary role. In our model, the RA reaction H + C5H5 is the dominant neutral source of C5H6, while the CH + C4H6 pathway contributes only marginally under nominal assumptions due to the low modeled abundance of 1,3-butadiene (C4H6). The abundance of C4H6 remains highly uncertain, as it cannot be directly observed and must be inferred indirectly through chemical proxies whose detections are still debated. Our results suggest that the reaction H + C4H5, and more generally hydrogen-atom-driven radiative association processes under low-density interstellar conditions, may represent a key missing ingredient in current chemical networks. Thus, quantifying the efficiency of these reactions at low pressure is essential to further constrain the formation of C5H6 and related cyclic hydrocarbons.
More broadly, this study illustrates the strong sensitivity of cyclopentadiene formation to a limited number of chemical pathways that connect linear hydrocarbon chains to the first five-membered ring, even when only a small subset of the associated parameters remains poorly constrained. Resolving the remaining discrepancy between models and observations will therefore require dedicated experimental and theoretical investigations of both the ionic reaction C3H7+ + C2H2, and neutral radiative association processes involving C4H5 and C5H5. Improving these constraints is a necessary step toward a coherent description of molecular mass growth from acyclic species to aromatic rings and, ultimately, to larger PAHs and carbonaceous nanostructures in cold interstellar environments.
Acknowledgements
The work was funded by the French “Agence Nationale de la Recherche” (ANR) under grant no. ANR-22-CE29-0013 (Project iSELECTION). This work was also supported by the Programme National “Physique et Chimie du Milieu Interstellaire” (PCMI) and a research grant (VIL71404) from VILLUM FONDEN.
References
- Agúndez, M., Cabezas, C., Marcelino, N., et al. 2025, A&A, 697, A82 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Anicich, V. G. 2003, JPL Publication-03-19, Pasadena, CA, USA [Google Scholar]
- Anicich, V. G., Wilson, P., & McEwan, M. J. 2003, JACSMAS, 14, 900 [Google Scholar]
- Anicich, V. G., Wilson, P. F., & McEwan, M. J. 2006, JACSMS, 17, 544 [Google Scholar]
- Bacskay, G. B., & Mackie, J. C. 2001, Phys. Chem. Chem. Phys., 3, 2467 [Google Scholar]
- Bouwman, J., Goulay, F., Leone, S. R., & Wilson, K. R. 2012, J. Phys. Chem. A, 116, 3907 [Google Scholar]
- Byrne, A. N., Xue, C., Van Voorhis, T., & McGuire, B. A. 2024, PCCP, 26, 26734 [Google Scholar]
- Cabezas, C., Agúndez, M., Pérez, C., et al. 2025, A&A, 701, L8 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Capron, M., Bourgalais, J., Abhinavam Kailasanathan, R. K., et al. 2015, PCCP, 17, 23833 [Google Scholar]
- Caster, K. L., Selby, T. M., Osborn, D. L., Le Picard, S. D., & Goulay, F. 2021, J. Phys. Chem. A, 125, 6927 [NASA ADS] [CrossRef] [Google Scholar]
- Cernicharo, J., Agúndez, M., Cabezas, C., et al. 2021a, A&A, 649, L15 [EDP Sciences] [Google Scholar]
- Cernicharo, J., Agúndez, M., Kaiser, R. I., et al. 2021b, A&A, 652, L9 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Cernicharo, J., Fuentetaja, R., Agúndez, M., et al. 2022, A&A, 663, L9 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Cernicharo, J., Cabezas, C., Fuentetaja, R., et al. 2024, A&A, 690, L13 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Chastaing, D., James, P., Sims, I., & Smith, I. 1999, PCCP, 1, 2247 [Google Scholar]
- Chin, C.-H., Chen, W.-K., Huang, W.-J., Lin, Y.-C., & Lee, S.-H. 2013, Icarus, 222, 254 [Google Scholar]
- Cooke, I. R., Xue, C., Changala, P. B., et al. 2023, APJ, 948, 133 [Google Scholar]
- Coutens, A., Loison, J.-C., Boulanger, A., et al. 2022, A&A, 660, L6 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Cunha De Miranda, B., Romanzin, C., Chefdeville, S., et al. 2015, JPCA, 119, 6082 [Google Scholar]
- Daugey, N., Caubet, P., Retail, B., et al. 2005, PCCP, 7, 2921 [NASA ADS] [CrossRef] [Google Scholar]
- Dobrijevic, M., Hébrard, E., Loison, J., & Hickson, K. 2014, Icarus, 228, 324 [NASA ADS] [CrossRef] [Google Scholar]
- Dobrijevic, M., Loison, J., Hickson, K., & Gronoff, G. 2016, Icarus, 268, 313 [NASA ADS] [CrossRef] [Google Scholar]
- Frank, P., Herzler, J., Just, T., & Wahl, C. 1994, in Proceedings of the Combustion Institute, 25 (Elsevier), 833 [Google Scholar]
- Frisch, M. J., Trucks, G. W., Schlegel, H. B., et al. 2016, Gaussian~16 Revision C.01, Gaussian Inc., Wallingford, CT [Google Scholar]
- Fuchs, V. R. 1961, Z. Naturforsch., 16a, 1026 [Google Scholar]
- Goettl, S. J., He, C., Paul, D., et al. 2022, J. Phys. Chem. A 126, 1889 [Google Scholar]
- Hahndorf, I., Lee, H. Y., Mebel, A. M., et al. 2000, J. Chem. Phys., 113, 9622 [CrossRef] [Google Scholar]
- Haider, N., & Husain, D. 1993, Int. J. Chem. Kinet., 25, 423 [Google Scholar]
- Hamberg, M., Vigren, E., Thomas, R., et al. 2011, EAS, 46, 241 [Google Scholar]
- Harding, L. B., Klippenstein, S. J., & Georgievskii, Y. 2007, JPCA, 111, 3789 [Google Scholar]
- He, C., Zhao, L., Doddipatla, S., et al. 2020, ChemPhysChem, 21, 1295 [Google Scholar]
- Hébrard, E., Dobrijevic, M., Loison, J.-C., Bergeat, A., & Hickson, K. 2012, A&A, 541, A21 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Hébrard, E., Dobrijevic, M., Loison, J.-C., et al. 2013, A&A, 552, A132 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Hickson, K. M., Loison, J.-C., & Wakelam, V. 2021, ACS Earth Space Chem., 5, 824 [NASA ADS] [CrossRef] [Google Scholar]
- Hickson, K. M., Loison, J.-C., & Wakelam, V. 2024, ACS Earth Space Chem., 8, 1087 [Google Scholar]
- Hincelin, U., Wakelam, V., Hersant, F., et al. 2011, A&A, 530, A61 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Hincelin, U., Commerçon, B., Wakelam, V., et al. 2016, APJ, 822, 12 [Google Scholar]
- Husain, D., & Ioannou, A. X. 1997, J. Chem. Soc. Faraday Trans., 93, 3625 [Google Scholar]
- Knyazev, V. D., & Slagle, I. R. 2001, J. Phys. Chem. A, 105, 3196 [Google Scholar]
- Lee, H.-Y., Kislov, V. V., Lin, S.-H., Mebel, A. M., & Neumark, D. M. 2003, Chem. Eur. J., 9, 726 [Google Scholar]
- Lifshitz, C., Gleitman, Y., Gefen, S., Shainok, U., & Dotan, I. 1981, IJMS, 40, 1 [Google Scholar]
- Loison, J. C., & Bergeat, A. 2004, Phys. Chem. Chem. Phys., 6, 5396 [Google Scholar]
- Loison, J.-C., & Bergeat, A. 2009, Phys. Chem. Chem. Phys., 11, 655 [Google Scholar]
- Loison, J.-C., Wakelam, V., Hickson, K. M., Bergeat, A., & Mereau, R. 2013, MNRAS, 437, 930 [NASA ADS] [CrossRef] [Google Scholar]
- Loison, J.-C., Wakelam, V., & Hickson, K. M. 2014, MNRAS, 443, 398 [Google Scholar]
- Loison, J., Hébrard, E., Dobrijevic, M., et al. 2015, Icarus, 247, 218 [CrossRef] [Google Scholar]
- Loison, J.-C., Agúndez, M., Marcelino, N., et al. 2016, MNRAS, 456, 4101 [Google Scholar]
- Loison, J.-C., Agúndez, M., Wakelam, V., et al. 2017, MNRAS, 470, 4075 [Google Scholar]
- Loison, J., Dobrijevic, M., & Hickson, K. 2019, Icarus, 329, 55 [CrossRef] [Google Scholar]
- Loison, J.-C., Rossi, C., Solem, N., et al. 2026, Nat. Astron., under review, [arXiv:2506.13290] [Google Scholar]
- Loru, D., Cabezas, C., Cernicharo, J., Schnell, M., & Steber, A. L. 2023, A&A, 677, A166 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Mallo, M., Agúndez, M., Cernicharo, J., & Molpeceres, G. 2025, A&A, 704, A249 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Manigand, S., Coutens, A., Loison, J.-C., et al. 2021, A&A, 645, A53 [EDP Sciences] [Google Scholar]
- McCarthy, M. C., Lee, K. L. K., Loomis, R. A., et al. 2021, Nat. Astron., 5, 176 [Google Scholar]
- McEwan, Murray, J., Scott, Graham, B. I., Adams, Nigel, G., et al. 1999, ApJ, 513, 287 [CrossRef] [Google Scholar]
- McEwan, M. J., & Anicich, V. G. 2007, Mass Spectrom. Rev., 26, 281 [Google Scholar]
- McEwan, M. J., Scott, G. B., & Anicich, V. G. 1998, IJMS, 172, 209 [Google Scholar]
- McGuire, B. A., Burkhardt, A. M., Kalenskii, S., et al. 2018, Science, 359, 202 [Google Scholar]
- McGuire, B. A., Loomis, R. A., Burkhardt, A. M., et al. 2021, Science, 371, 1265 [Google Scholar]
- Mu, X., Lu, I. C., Lee, S.-H., Wang, X., & Yang, X. 2004, J. Chem. Phys., 121, 4684 [Google Scholar]
- Newby, J. J., Stearns, J. A., Liu, C.-P., & Zwier, T. S. 2007, J. Phys. Chem. A, 111, 10914 [Google Scholar]
- Petrie, S., Javahery, G., & Bohme, D. K. 1992, J. Am. Chem. Soc., 114, 9205 [Google Scholar]
- Rossi, C., Alcaraz, C., Thissen, R., & Jacovella, U. 2023, J. Phys. Org. Chem., 36, e4489 [Google Scholar]
- Rossi, C., Muller, G., Solem, N., et al. 2025, ACS Earth Space Chem. 9, 349 [Google Scholar]
- Roy, K., Horn, C., Frank, P., Slutsky, V. G., & Just, T. 1998, Symp. (Int.) Combust., 27, 329 [Google Scholar]
- Ruaud, M., Wakelam, V., & Hersant, F. 2016, MNRAS, 459, 3756 [Google Scholar]
- Scott, G. B. I., Milligan, D. B., Fairley, D. A., Freeman, C. G., & McEwan, M. J. 2000, J. Chem. Phys., 112, 4959 [Google Scholar]
- Sita, M. L., Changala, P. B., Xue, C., et al. 2022, Astrophys. J., 938, L12 [Google Scholar]
- Smith, R. D., & Futrell, J. H. 1978, Int. J. Mass Spectrom. Ion Phys., 26, 111 [Google Scholar]
- Smyth, K. C., G., L. S., & Ausloos, P. 1982, Combust. Sci. Technol., 28, 147 [CrossRef] [Google Scholar]
- Snow, T. P., Le Page, V., Keheyan, Y., & Bierbaum, V. M. 1998, Nature, 391, 259 [Google Scholar]
- Suutarinen, A., Geppert, W. D., Harju, J., et al. 2011, A&A, 531, A121 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Thaddeus, P., Vrtilek, J., & Gottlieb, C. 1985, Astrophys. J., 299, L63 [Google Scholar]
- Vuitton, V., Yelle, R., Lavvas, P., & Klippenstein, S. 2011, APJ, 744, 11 [Google Scholar]
- Wakelam, V., Ruaud, M., Gratier, P., & Bonnell, I. 2019, MNRAS, 486, 4198 [NASA ADS] [CrossRef] [Google Scholar]
- Wakelam, V., Dartois, E., Chabot, M., et al. 2021, A&A, 652, A63 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Wakelam, V., Gratier, P., Loison, J.-C., et al. 2024, A&A, 689, A63 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Wenzel, G., Cooke, I. R., Changala, P. B., et al. 2024, Science, 386, 810 [NASA ADS] [CrossRef] [Google Scholar]
- Wenzel, G., Gong, S., Xue, C., et al. 2025, Astrophys. J., 984, L36 [Google Scholar]
- Yang, Z., Medvedkov, I. A., Goettl, S. J., et al. 2024, PNAS, 121, e2409933121 [Google Scholar]
- Zhao, Y., & Truhlar, D. G. 2008, Theor. Chem. Acc., 120, 215 [CrossRef] [Google Scholar]
- Zhou, F., Ma, S., Li, L., et al. 2025, JCP, 162, 9 [Google Scholar]
Appendix A Elemental abundances and other model parameters
Elemental abundances and other model parameters
Appendix B C2H4+ + C3H4 reactions including secondary products
 |
Fig. B.1 Panels a and c display the C2H4+ parent-ion signal from the photoionization of ethylene. Panels b and d show the reaction cross sections (left axis) and rate constants (right axis) with propyne and allene, respectively, as a function of photon energy. |
Appendix C Chemical network used for the model
Chemical network used for the model. Rate coefficients α(T/300)βexp(-γ/T) are in units of cm3 s−1. ΔHR in kJ mol−1. Reactions highlighted in grey such as 15 are not included in the network.
All Tables
Chemical network used for the model. Rate coefficients α(T/300)βexp(-γ/T) are in units of cm3 s−1. ΔHR in kJ mol−1. Reactions highlighted in grey such as 15 are not included in the network.
All Figures
 |
Fig. 1 Panels a and c display the C2H4+ parent-ion signal from the photoionization of ethylene. Panels b and d show the reaction cross sections (left axis) and rate constants (right axis) with propyne and allene, respectively, as a function of photon energy. |
| In the text | |
 |
Fig. 2 Schematic diagram showing the main formation routes of C5H6 in TMC-1. Percentages written in bold indicate the contribution of the reactions in the nominal model. Percentages in normal text, associated linked with dashed arrows, correspond to their contribution if the C4H5 + H reaction is included. |
| In the text | |
 |
Fig. 3 Gas–grain astrochemical model results for C5H6 in dark clouds as a function of cloud age. (a–c) Nominal network results (solid black line); nominal network but with only some specific reactions as a source of C5H6 (solid red line). (d) Nominal with H + C4H5 leading to C4H6 (solid black line), nominal with H + C4H5 leading to C4H6 with only H + C5H5 leading to C5H6 (solid red line), and nominal with H + C4H5 leading to C4H6 with only CH + C4H6 leading to C5H6 (solid blue line). The horizontal gray region represents the observed abundances in TMC-1 with an arbitrary error associated (±2). The estimated age of the cloud, marked by a vertical gray region, corresponds to the time at which our model predictions best match the observed abundances of 67 key species. |
| In the text | |
 |
Fig. B.1 Panels a and c display the C2H4+ parent-ion signal from the photoionization of ethylene. Panels b and d show the reaction cross sections (left axis) and rate constants (right axis) with propyne and allene, respectively, as a function of photon energy. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


