Fig. 4
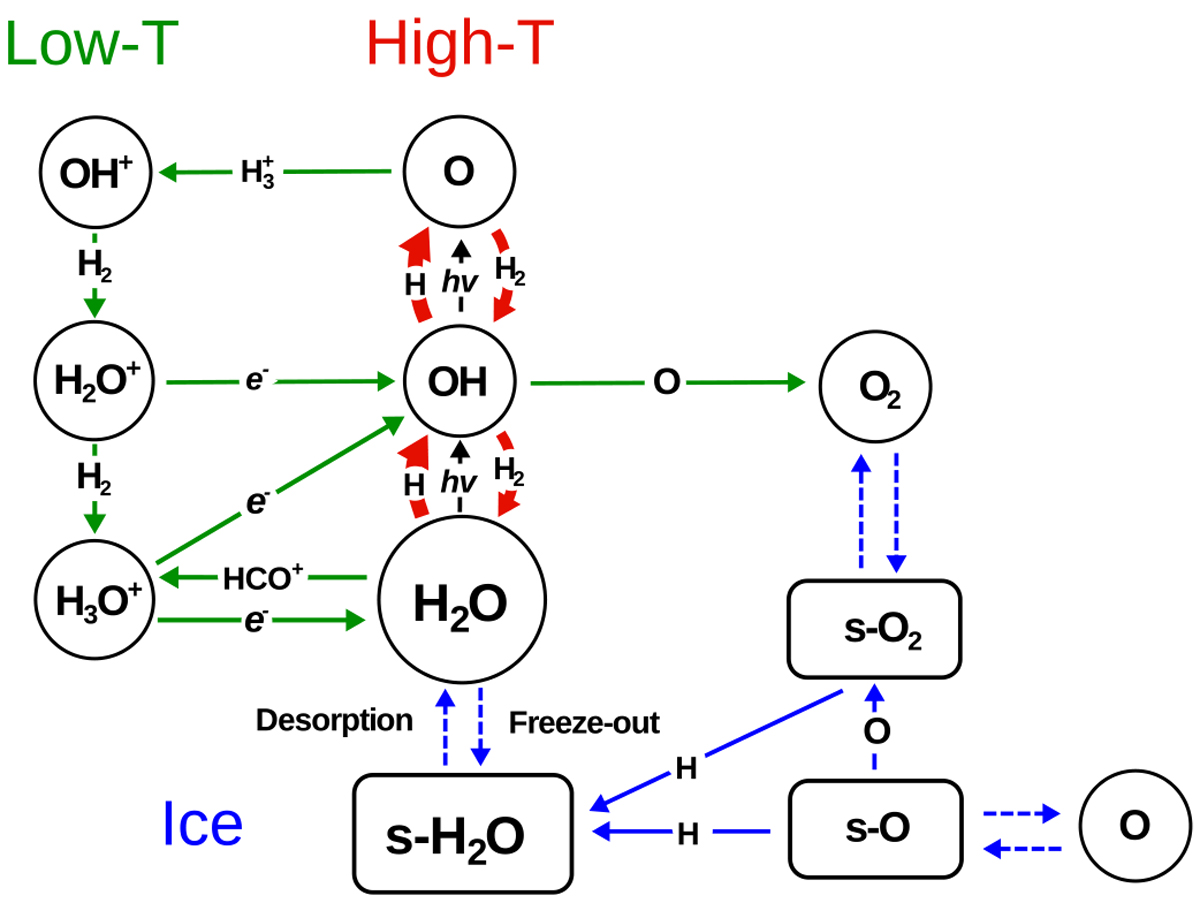
Simplified gas-phase and solid-state reaction network leading to the formation and destruction of H2O. s-X denotes species X on the ice surface. Three routes to water can be distinguished: (i) ion-molecule gas-phase chemistry which dominates H2O formation atlow temperatures and low densities (green); (ii) high-temperature neutral-neutral chemistry which is effective above ~250 K when energy barriers can be overcome (red); and (iii) solid-state chemistry (blue). The full solid-state network is presented in Lamberts et al. (2013). Chemically related molecules discussed in this paper such as OH, O2 and HCO+ are shown as well.
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


