A&A 492, 719-724 (2008)
DOI: 10.1051/0004-6361:200810536
J. B. Bossa - P. Theulé - F. Duvernay - F. Borget - T. Chiavassa
Université de Provence, Laboratoire de Physique des Interactions Ioniques et Moléculaires Centre de St-Jérôme, Avenue Escadrille Normandie-Niémen, 13397 Marseille, France
Received 7 July 2008 / Accepted 3 October 2008
Abstract
Context. We study carbamic acid [NH2COOH] and ammonium carbamate [NH2COO-][NH4+] formation in interstellar ice analogs.
Aims. We demonstrate how carbamic acid [NH2COOH] and ammonium carbamate [NH2COO-][NH4+] can be formed from both thermal reactions and energetic photons in an NH3:CO2 ice mixture.
Methods. Infrared and mass spectroscopy are used to monitor NH3:CO2 ice mixture evolution during both warming and VUV photon irradiation.
Results. Carbamic acid and ammonium carbamate can be produced thermally in a 1:1 ratio from NH3 and CO2 above 80 K. They can be also formed in a 28:1 ratio by less efficient processes such as energetic photons. Our study and its results provide fresh insight into carbamic acid formation in interstellar ices.
Conclusions. We demonstrate that care is required to separate irradiation-induced reactivity from purely thermal reactivity in ices in which ammonia and carbon dioxide are both present. From an interstellar chemistry point of view, carbamic acid and ammonium carbamate are readily produced from the ice mantle of a typical interstellar grain and should therefore be a detectable species in molecular clouds.
Key words: astrochemistry - ISM: molecules - methods: laboratory
In molecular clouds, interstellar grains are responsible for the synthesis of molecules that cannot be formed in the gas phase of the interstellar medium (ISM). Observations from the InfraRed Astronomical Satellite (IRAS), Infrared Space Observatory (ISO), and Spitzer missions provide a wealth of spectral information in the mid infrared (MIR) range and enable the identification of several simple species frozen in interstellar grains (van Dishoeck 2004). More complex products can be formed by thermal processes from simpler species (Schutte et al. 1993a,b). The grain surface offers a place where two species can meet and possibly react. This surface can also have a catalytic role because it can enhance the reaction by lowering the activation barrier (Duvernay et al. 2005). These grains are also subjected to non-thermal energetic processes through different types of bombardment (cosmic rays, electrons, VUV radiation). These radiations can form radicals and ions that can react with no or little activation barrier. The complex species produced can be released into the gas phase of the ISM and in the case of star-forming regions, eventually be incorporated into comets and meteorites. A large amount of laboratory and observational data are contributing to a a more robust understanding of complex molecule formation in the solid phase of the ISM (Schutte 1995; Elsila et al. 2007; Greenberg 2002).
We focus our study on carbamic acid NH2COOH, since it is the simplest molecule that has both an amino group and a carboxylic group, and may be an intermediate (or a deadlock) in the route toward the formation of the simplest amino acid, glycine NH2CH2COOH. Carbamic acid has been observed neither in the gas phase nor in the solid phase of ISM. Carbamic acid is not stable at room temperature in gas phase and spontaneously dissociates into NH3 and CO2 (Aresta et al. 2000; Remko et al. 1995). Therefore, there exists no gas phase spectroscopic studies of this molecule in the lab or in space. If it is present in the ISM, such as in the cold environment of outer planetary objects, including comets, carbamic acid must have be formed and be stored in a stable form such as a salt (ammonium carbamate) or a multimer, in a solid icy phase.
To produce carbamic acid, we start from a binary mixture of
NH3 and CO2, since these two species are known to exist
in interstellar grains (Whittet et al. 1996). Ammonia can be identified
by its bands at 3367 cm-1 (2.97 ![]() m), and at 1069.5 cm-1 (9.35
m), and at 1069.5 cm-1 (9.35 ![]() m) (Dartois 2004). The ubiquitous CO2is identified by its bending mode band at 667 cm-1 (15.2
m) (Dartois 2004). The ubiquitous CO2is identified by its bending mode band at 667 cm-1 (15.2 ![]() m) and its asymmetric stretching mode at 2349 cm-1 (4.27
m) and its asymmetric stretching mode at 2349 cm-1 (4.27 ![]() m ) (Dartois 2004). Their abundances are estimated to be
less than 5-10% (Dartois 2004), (Lacy et al. 1998) and 10-23% (Gerakines et al. 1995) of the abundance of the H2O ice, respectively.
m ) (Dartois 2004). Their abundances are estimated to be
less than 5-10% (Dartois 2004), (Lacy et al. 1998) and 10-23% (Gerakines et al. 1995) of the abundance of the H2O ice, respectively.
Low temperature reactivity of thin solid films formed by condensation of the gas phase NH3 and CO2 was investigated by Frasco (1964). He reported that NH3 and CO2 react to form ammonium carbamate [NH2COO-][NH4+] under two solid forms, that he called amorphous and crystalline. On the same NH3:CO2system, Hisatsune (1984) assigned the bands previously related to the crystalline form by Frasco (1964), to a prereactive complex (NH3)2CO2. These two studies clearly demonstrated that ammonium carbamate [NH2COO-][NH4+] is formed easily in these binary NH3:CO2 ice mixtures, although they did not provide evidence of the formation of neutral carbamic acid NH2COOH. However, there was controversy over the assignment of some vibrational bands to different types of amorphous ammonium carbamate solid.
Non-thermal processing of NH3:CO2 and NH3:CO2:H2O mixtures have also been studied. (Khanna & Moore 1999) bombarded NH3/(H2O+CO2) ice mixtures (a layer of NH3 covered by a layer of a H2O+CO2 ice) with 1-MeV protons. After bombardment, the samples had been warmed to 250 K and were monitored by IR spectroscopy. At 250 K, the remaining organic residue was assigned to carbamic acid in its zwitterionic form NH3+COO-. Chen et al. (2007) performed irradiation of NH3:CO2:H2O ice mixtures at 15 K with UV/EUV photons of 4-20 eV (310-62 nm), and followed the reaction by IR and mass spectroscopy, while warming the ice films to 250 K. Along with the IR features HNCO, OCN-, NH4+, CO and NH2(CO)H, they identified carbamic acid in its neutral form. It is important to note that in every study, carbamic acid was identified after both irradiation and heating to 250 K. We will show that heating to 250 K after irradiation at low temperature has for effect to allow the reactants that have not been decomposed by irradiation to thermally react. Results from this study indicate that the identification in this type of ices must be considered with care to be able to separate irradiation induced reactivity from thermal induced reactivity.
In the present work, we study the thermal reactivity of NH3:CO2 ices. We demonstrate that NH3:CO2 ice mixtures react above 80 K to form both ammonium carbamate ([NH2COO-][NH4+]) and carbamic acid dimers (NH2COOH)2. In a second type of experiments, we study the photon-induced reactivity by irradiating the NH3:CO2mixture with VUV photons at 10 K, followed by warming at room temperature.
Carbon dioxide (CO2) and ammonia (NH3) are commercially
available in the form of 99.9995% pure gas from Linde and Air
Liquide. The NH3:CO2 gas mixtures were prepared into a
primary vacuum pumped mixing line (10-3 mbar) at room
temperature. They were then deposited onto a gold plated metal
surface cooled down to 10 K by a Model 21 CTI cold head within a
high vacuum chamber ca 10-7 mbar). Typical deposition times
were between 5 and 10 min. The mixing line was heated by a
heatgun under vacuum conditions between two different experiments
to avoid contamination of our system. Pure NH3, pure CO2, and
NH3:CO2 were deposited onto the cold surface. The IR
spectra are recorded between 4000 cm-1 and 650 cm-1 in
reflexion mode onto the gold plated surface using a NICOLET Magna
750 FTIR spectrometer. A typical spectrum has a 1 cm-1resolution and is averaged over one hundred interferograms. The
sample was warmed and its temperature controlled by a Lakeshore
Model 320 temperature controller. The temperature ramp rate was
set to 4 K min-1, and the IR spectrum was monitored during
the temperature ramp. Mass spectra were recorded using a MKS
Microvision-IP plus RGA quadrupole mass spectrometer as the
products were desorbed during the temperature ramp. The mass
spectra were recorded between 1 and 80 amu (atomic mass units).
The ionization source was an 70 eV impact electronic source.
The IR and mass analysis were not performed simultaneously because of the limitations of our experimental configuration; as a consequence, we repeated each experiment to be able to
acquire both the IR spectra and the mass spectra. The VUV
radiation (
![]() nm) was generated by a microwave
discharge hydrogen flow lamp (Opthos instruments). The flux of
photons from the hydrogen lamp was estimated to be about 1015 cm-2 s-1.
nm) was generated by a microwave
discharge hydrogen flow lamp (Opthos instruments). The flux of
photons from the hydrogen lamp was estimated to be about 1015 cm-2 s-1.
The true concentration ratio was obtained using two methods:
standard manometric techniques and by using the IR spectra to
estimate the column density of CO2 and NH3 according to
their band strengths. However, the ratio calculated using the
second method depends on the band strengths provided by the
literature. The band strength for the CO2 asymmetric stretching
band at 2340 cm-1 was measured to be
![]() cm molecule-1 for the pure solid
(Gerakines et al. 1995; Yamada & Person 1964), while in water ice a value of
cm molecule-1 for the pure solid
(Gerakines et al. 1995; Yamada & Person 1964), while in water ice a value of
![]() cm molecule-1 was found
(Sandford & Allamandola 1990). The band strength for the wagging mode of
NH3 at 1070 cm-1 varies from
cm molecule-1 was found
(Sandford & Allamandola 1990). The band strength for the wagging mode of
NH3 at 1070 cm-1 varies from
![]() to
to
![]() cm molecule-1(Kerkhof et al. 1999; d'Hendecourt et al. 1986) in its pure solid form. The values
of the band strengths for these two molecules depend on the
nature, composition, and temperature of the ice in which they are
found. We therefore used the values given by Yamada & Person
(1964) for CO2 (
cm molecule-1(Kerkhof et al. 1999; d'Hendecourt et al. 1986) in its pure solid form. The values
of the band strengths for these two molecules depend on the
nature, composition, and temperature of the ice in which they are
found. We therefore used the values given by Yamada & Person
(1964) for CO2 (
![]() cm molecule-1) and
the value given by Kerkhof et al. (1999) for the NH3(
cm molecule-1) and
the value given by Kerkhof et al. (1999) for the NH3(
![]() cm molecule-1) (Kerkhof et al. 1999) with
these values, we derived a ratio of 1.05 which is consistent with
the value that we mesured using mamonetric techniques. The
thickness of the deposited solid films, assuming densities for
CO2 of 1.60 g cm-3 and for NH3 of 0.69 g cm-3,
are estimated to be around 0.1
cm molecule-1) (Kerkhof et al. 1999) with
these values, we derived a ratio of 1.05 which is consistent with
the value that we mesured using mamonetric techniques. The
thickness of the deposited solid films, assuming densities for
CO2 of 1.60 g cm-3 and for NH3 of 0.69 g cm-3,
are estimated to be around 0.1 ![]() m (Bennett et al. 2004). We can
also monitor the NH3 and CO2 IR band intensities as they
are consumed during the reaction, and estimate how much of each
product is formed as well as the band strength of some of their
bands.
m (Bennett et al. 2004). We can
also monitor the NH3 and CO2 IR band intensities as they
are consumed during the reaction, and estimate how much of each
product is formed as well as the band strength of some of their
bands.
Pure NH3 and CO2 solids are deposited independently at 10 K and their respective IR spectra are displayed in Fig. 1. The assignment of the bands of these species is well known and reported in Table 1 in pure solids and in the 1:1 mixture. In the pure solid, at a rate of 4 K min-1CO2 starts to sublime at 90 K, which is consistent with the results of a previous study by Sandford & Allamandola (1990). Based on the IR spectra CO2 fully disappears at 105 K. The sublimation of pure NH3 commences at 100 K (Sandford & Allamandola 1993) and NH3 fully disappears at 120 K.
![\begin{figure}
\par\includegraphics[width=8.5cm,clip]{1053fig1.eps}
\end{figure}](/articles/aa/full/2008/48/aa10536-08/Timg9.gif) |
Figure 1: Infrared spectra recorded at 10 K a) of pure NH3, b) CO2 ices, and c) a NH3:CO2 binary mixture in a 1:1 ratio. Arrow: NH stretching mode relative to the NH3:CO2 complex. |
| Open with DEXTER | |
Table 1: Infrared absorption bands and assignments of NH3 and CO2, in pure solids and in NH3:CO2 ice mixtures at 10 K.
A binary (NH3:CO2) ice mixture is then deposited at 10 K
within a 1:1 ratio. No reaction occurs at this temperature and
the IR spectrum of the mixture is mainly the superposition of the
NH3 and CO2 spectra as seen in Fig. 1c.
Nevertheless, an extra feature can be observed in the ![]() NH
stretching region in the 1:1 spectrum at 3410 cm-1. This
frequency is closed to the observed value in its gas phase at 3444 cm-1 for the
NH
stretching region in the 1:1 spectrum at 3410 cm-1. This
frequency is closed to the observed value in its gas phase at 3444 cm-1 for the ![]() NH mode of NH3 (Jacox et al. 1963). This mode is indeed especially sensitive to the hydrogen bonding. The
gas phase frequency is lowered from 3444 cm-1 to 3378 cm-1 in crystalline NH3, and to 3404 cm-1 for NH3 dimer in a N2 matrix (Pimentel et al. 1962). In CO2matrices with mole ratios CO2:NH3 ranging from 100 to
700, this mode was observed at 3434 cm-1 (Jacox et al. 1963). This
new feature is consistent with the presence of a T-shaped
NH3:CO2 molecular complex in which the nitrogen atom bonds
with the carbon of CO2. The small frequency shift that we
observe between the
NH mode of NH3 (Jacox et al. 1963). This mode is indeed especially sensitive to the hydrogen bonding. The
gas phase frequency is lowered from 3444 cm-1 to 3378 cm-1 in crystalline NH3, and to 3404 cm-1 for NH3 dimer in a N2 matrix (Pimentel et al. 1962). In CO2matrices with mole ratios CO2:NH3 ranging from 100 to
700, this mode was observed at 3434 cm-1 (Jacox et al. 1963). This
new feature is consistent with the presence of a T-shaped
NH3:CO2 molecular complex in which the nitrogen atom bonds
with the carbon of CO2. The small frequency shift that we
observe between the ![]() NH of free and complexed ammonia (30 cm-1) excludes the possibility of any hydrogen bond between hydrogen of NH3 and CO2. This T-shaped structure has been
characterized in the gas phase (Fraser et al. 1984) and by quantum
calculation (Jönsson et al. 1977).
NH of free and complexed ammonia (30 cm-1) excludes the possibility of any hydrogen bond between hydrogen of NH3 and CO2. This T-shaped structure has been
characterized in the gas phase (Fraser et al. 1984) and by quantum
calculation (Jönsson et al. 1977).
In a first series of experiments, the ice mixture were warmed to room temperature, to take into account the thermal reactivity alone. In a second series of experiments, the ice mixture are irradiated by VUV photons at 10 K followed by warming to room temperature.
A binary NH3:CO2=1:1 ice was deposited at 10 K. The IR spectrum evolution is shown in Fig. 2. No reaction occurs at 10 K. When the sample is warmed to above 80 K, NH3, and CO2 are consumed, and new features are observed. After complete sublimation of the reactants around 130 K, two sets of bands can be isolated. One, labelled C decreases more rapidly than the other labelled D, as the temperature ramp continues. The two set of bands presumably correspond to two different species, the C species being the main product at 140 K. They both sublimate in the range 230-260 K. The sublimation was monitored by both FTIR and mass spectroscopy. The bands of these two species were separated by taking a difference spectrum between 230 K and 140 K as shown in Fig. 3.
![\begin{figure}
\par\includegraphics[width=9cm,clip]{1053fig2.eps}
\end{figure}](/articles/aa/full/2008/48/aa10536-08/Timg23.gif) |
Figure 2: Infrared spectrum of the thermal evolution of a NH3:CO2=1:1 binary ice mixture, a) at 10 K, b) at 90 K, c) at 140 K, and d) at 230 K. |
| Open with DEXTER | |
![\begin{figure}
\par\includegraphics[width=8.8cm,clip]{1053fig3.eps}
\end{figure}](/articles/aa/full/2008/48/aa10536-08/Timg24.gif) |
Figure 3:
Infrared spectra of a) carbamic acid dimer (NH2COOH)2 (labelled D in the text), obtained by substracting the spectrum of
NH3:CO2 at 230 K and at 140 K and b) ammonium carbamate
[NH2COO-][NH4+] (labelled C in the text),
obtained by substracting the spectrum of the dimer on the spectrum
of NH3:CO2 at 230 K. Upper panel: the evolution of the peak
at m/z=61 and m/z=44, relative to NH2COOH
|
| Open with DEXTER | |
Table 2: Infrared absorption bands and assignments of the products formed from a binary NH3:CO2=1:1.
The C species is assigned to ammonium carbamate
[NH2COO-][NH4+] following the assignment
given by Frasco (1964) and Hisatsune (1984). The absorption bands
of the spectrum for the C species recorded at 140 K is shown in
Fig. 3b, and their frequencies are reported in Table 2 along with their assignments. The IR spectum of ammonium carbamate is dominated by
![]() COO- at 1553 cm-1. There is controversy about the bands at 1394 cm-1and 1117 cm-1 attributed to
COO- at 1553 cm-1. There is controversy about the bands at 1394 cm-1and 1117 cm-1 attributed to ![]() CN stretch and a
symmetric COO- stretch by Frasco
and inversely by Hisatsune. Following the convention of Bossa et al. (2008) work, we assigned the bands at 1394 and 1117 cm-1 to
CN stretch and a
symmetric COO- stretch by Frasco
and inversely by Hisatsune. Following the convention of Bossa et al. (2008) work, we assigned the bands at 1394 and 1117 cm-1 to
![]() COO- and
COO- and ![]() CN, respectively. In this
we also observed
CN, respectively. In this
we also observed
![]() COO- stretching mode of
methylammonium methylcarbamate [CH3NHCOO-][CH3NH3+] at 1409 cm-1and
COO- stretching mode of
methylammonium methylcarbamate [CH3NHCOO-][CH3NH3+] at 1409 cm-1and ![]() CN at 1141 cm-1.
CN at 1141 cm-1.
The D species, which produces the difference spectrum between 230 K and 140 K (Fig. 3a), is assigned to the dimer of the neutral carbamic acid NH2COOH. This is consistent with our
previous study of the thermal reaction between methylamine
(CH3NH2) and carbon dioxide (CO2) in which we
observed the formation of the centro symmetric dimer of
methylcarbamic acid (Bossa et al. 2008). Jamróz et al. (2002)
calculated that the dimer of carbamic acid should be stabilized by
ca 90 kJ mol-1. The band at 3462 cm-1 is typical of a
![]() NH stretch and the band at 3140 cm-1 of the associated
OH stretch. Since the OH stretch of one monomer participates in a
hydrogen bond with the carbonyl of the other monomer, the OH band
is broadened considerably and shifted with respect to a free OH
oscillator. The
NH stretch and the band at 3140 cm-1 of the associated
OH stretch. Since the OH stretch of one monomer participates in a
hydrogen bond with the carbonyl of the other monomer, the OH band
is broadened considerably and shifted with respect to a free OH
oscillator. The ![]() C=O stretching mode is observed at 1691 cm-1, which is also typical of dimer-like carboxylic acid. The infrared observations are correlated with mass spectrometry
experiments during the warming of the sample to room temperature
(Fig. 3). We observe three clear peaks for m/z=44(CO2) at 104 K, 230 K, and 260 K, which are correlated with
the observed loss of non-reacting CO2, carbamate, and
carbamic acid, respectively. For m/z=61, we observed only one peak
at 260 K, corresponding to the molecular ion peak of carbamic acid
(NH2COOH
C=O stretching mode is observed at 1691 cm-1, which is also typical of dimer-like carboxylic acid. The infrared observations are correlated with mass spectrometry
experiments during the warming of the sample to room temperature
(Fig. 3). We observe three clear peaks for m/z=44(CO2) at 104 K, 230 K, and 260 K, which are correlated with
the observed loss of non-reacting CO2, carbamate, and
carbamic acid, respectively. For m/z=61, we observed only one peak
at 260 K, corresponding to the molecular ion peak of carbamic acid
(NH2COOH
![]() ). We can therefore deduce from these
results that (i) at 230 K, ammonium carbamate decomposes
spontaneously into CO2 and NH3; (ii) at 260 K,
when carbamic acid is desorbing, it partially decomposes into
CO2 and NH3. In its gas phase, neutral carbamic acid is
known to be unstable (Remko et al. 1995). The reactivity pathway for
solid NH3 and CO2 1:1 is summarized in Fig. 4.
The integrated band strength, A (cm molecule-1),
corresponding to the most typical bands of ammonium carbamate and
dimer of carbamic acid are determined by integrating the optical
depth
). We can therefore deduce from these
results that (i) at 230 K, ammonium carbamate decomposes
spontaneously into CO2 and NH3; (ii) at 260 K,
when carbamic acid is desorbing, it partially decomposes into
CO2 and NH3. In its gas phase, neutral carbamic acid is
known to be unstable (Remko et al. 1995). The reactivity pathway for
solid NH3 and CO2 1:1 is summarized in Fig. 4.
The integrated band strength, A (cm molecule-1),
corresponding to the most typical bands of ammonium carbamate and
dimer of carbamic acid are determined by integrating the optical
depth ![]() of the bands according to the equation:
of the bands according to the equation:
![]() and estimating the total column density
of carbamate and dimer of carbamic acid molecules formed N(molecule cm-2) from the amount of CO2 molecules having
reacted to produce the carbamate and carbamic acid. To evaluate
the amount of CO2 molecules, we used the C=O asymmetric
stretching mode of CO2 at 2337 cm-1 (
and estimating the total column density
of carbamate and dimer of carbamic acid molecules formed N(molecule cm-2) from the amount of CO2 molecules having
reacted to produce the carbamate and carbamic acid. To evaluate
the amount of CO2 molecules, we used the C=O asymmetric
stretching mode of CO2 at 2337 cm-1 (
![]() cm molecule-1) (Gerakines et al. 1995; Yamada & Person 1964). It is also clear
that all the observed loss of CO2 cannot be due only to the
thermal reactivity, but can be also caused by its sublimation
(Fig. 3). To evaluate the amount of non-reacting CO2that sublimates, we compare the relative surface area of desorbing
CO2 at 104 K and the area of both surfaces, corresponding to
carbamate and carbamic acid (230 K and 260 K, Fig. 3).
We estimate that 49% of the deposited CO2 sublimates without reacting with NH3. This implies that only 51% of the initial CO2 is involved in the thermal reaction.
cm molecule-1) (Gerakines et al. 1995; Yamada & Person 1964). It is also clear
that all the observed loss of CO2 cannot be due only to the
thermal reactivity, but can be also caused by its sublimation
(Fig. 3). To evaluate the amount of non-reacting CO2that sublimates, we compare the relative surface area of desorbing
CO2 at 104 K and the area of both surfaces, corresponding to
carbamate and carbamic acid (230 K and 260 K, Fig. 3).
We estimate that 49% of the deposited CO2 sublimates without reacting with NH3. This implies that only 51% of the initial CO2 is involved in the thermal reaction.
To evaluate the amount of ammonium carbamate formed, we used the
band strengths of methylammonium methylcarbamate for the
![]() COO- stretching mode (
COO- stretching mode (
![]() cm molecule-1) (Bossa et al. 2008). Thus, given of the total amount of CO2 molecules that reacted and the amount of ammonium
carbamate formed, we can deduce easily the amount of carbamic acid
formed by evaluating: N (CO2)
cm molecule-1) (Bossa et al. 2008). Thus, given of the total amount of CO2 molecules that reacted and the amount of ammonium
carbamate formed, we can deduce easily the amount of carbamic acid
formed by evaluating: N (CO2)
![]() = N (carbamate) +
N (carbamic acid). The ammonium carbamate and carbamic acid band strengths are listed
in Table 2. Finally, we estimate that the warming of
the 1:1 NH3:CO2 mixture at 140 K produces 50% of ammonium
carbamate, i.e. the carbamate:carbamic acid ratio is about 1:1.
= N (carbamate) +
N (carbamic acid). The ammonium carbamate and carbamic acid band strengths are listed
in Table 2. Finally, we estimate that the warming of
the 1:1 NH3:CO2 mixture at 140 K produces 50% of ammonium
carbamate, i.e. the carbamate:carbamic acid ratio is about 1:1.
![\begin{figure}
\par\includegraphics[width=9cm,clip]{1053fig4.eps}
\end{figure}](/articles/aa/full/2008/48/aa10536-08/Timg42.gif) |
Figure 4: Reactivity of solid NH3 and CO21:1. |
| Open with DEXTER | |
Binary NH3:CO2 = 1:1 ices were deposited at 10 K and
irradiated by VUV photons. After 720 min of irradiation, newly
formed species were observed (Fig. 5) and almost 50% of
the reactants had been consumed. Due to the VUV light flux which
used, this laboratory irradiation time was equivalent to 107 years in dense molecular clouds and this is consistent with the residence time of ice in dense molecular clouds (about 108 yrs) (Greenberg 1999). At 10 K no thermal reaction can occur, so
the observed reactivity was induced only by photon irradiation.
Species formed along with their characteristic bands are listed in
Table 3. After 720 min of irradiation, the
spectrum shows two strong bands at 2164 and 2140 cm-1,
assigned to OCN- (; Grim et al. 1987) and CO
(Sandford et al. 1988); (Gerakines et al. 1995), respectively. In the
carbonyl region (1800-1500 cm-1), we identify the dimer of
carbamic acid with the band at 1691 cm-1, and the ammonium
carbamate with the band at 1548 cm-1, which are the strongest
bands for these species. The other vibrational bands for these two
species are listed in Table 3. Besides carbamic
acid and ammonium carbamate formation, VUV irradiation of
NH3:CO2 ice produces an additional product: ammonium
formate [HCOO-][NH4+]. This species was identified
by means of its absorbtion bands at 1590 and 1389 cm-1(Schutte et al. 1999). The chemical evolution of the irradiated
NH3:CO2 ice is assumed to proceed along the following path:
direct dissociation of NH3 and CO2:
| (1) |
| (2) |
| (3) |
| (4) |
| (5) |
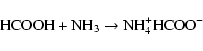 |
(6) |
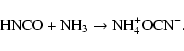 |
(7) |
| (8) |
| (9) |
![\begin{figure}
\par\includegraphics[width=8cm,clip]{1053fig5.eps}
\end{figure}](/articles/aa/full/2008/48/aa10536-08/Timg52.gif) |
Figure 5: Infrared spectra of a) carbamic acid dimer (NH2COOH)2, b) ammonium carbamate [NH2COO-][NH4+], c) ammonium formate [HCOO-][NH4+] at 80 K, and d) difference spectrum between the 720 min. and 0 min. VUV irradiation times at 10 K. |
| Open with DEXTER | |
We can see from Fig. 6b that NH3 and CO2 are still present even after 720 min of VUV irradiation. During warming the irradiated sample, the remaining NH3 and CO2 therefore react to produce ammonium carbamate and carbamic acid (as a dimer) as in the pure thermal reactivity case. Results from this study demonstrate that both carbamic acid and ammonium carbamate can be formed from both thermal and photochemical reaction. Nevertheless, the carbamate:carbamic acid ratio depends on the energetic process. In a pure thermal reaction, the carbamate:carbamic acid ratio is 1:1, while the VUV irradiation produces a 1:28 ratio. The interstellar detection of ammonium carbamate and carbamic acid and more specifically the ratio between them, could provide evidence of the origin of their formation in the interstellar medium.
![\begin{figure}
\par\includegraphics[width=8cm,clip]{1053fig6.eps}
\end{figure}](/articles/aa/full/2008/48/aa10536-08/Timg53.gif) |
Figure 6: Infrared spectra for the NH3:CO2=1:1 binary ice mixture a) at 10 K, b) after 720 min. VUV irradiation at 10 K, c) irradiated sample warming to 110 K, and d) warming to 230 K. |
| Open with DEXTER | |
Table 3: Infrared absorption bands of the species formed upon VUV irradiation of a NH3:CO2 ice at 10 K.
The comparison between our results and cryogenic matrix studies
indicate that binary NH3:CO2 ice mixtures at 10 K enable
molecular interactions to occur between NH3 and CO2molecules such as a T-shaped complex (Jacox et al. 1963). This
interaction foreshadows C-N bond formation. Warming the mixture
around 80 K gives ammonium carbamate [NH2COO-][NH4+] and neutral carbamic acid
NH2COOH in its dimer form D with a ratio of ![]() 1:1. Both
species desorb in the range 230-260 K. Many molecular clouds have
temperatures above 80 K and carbamic acid is therefore likely to
be present in the ISM, stored in its salt form, i.e. such as
ammonium carbamate [NH2COO-][NH4+] in the icy
mantle of the interstellar grains. This straightforward reaction
produces ammonium carbamate two of the most abundant species in
the ice mantle, NH3 and CO2. However, it is unlikely
that all the CO2 and NH3 present in interstellar grains will
be converted into ammonium carbamate and carbamic acid. In genuine
interstellar ices, a higher number of molecules are present in the
icy mantles, resulting in sophisticated and complex chemical
reaction network. The dilution in water ice should lower the
reaction rate. Competitive reaction, such as the thermal reaction
of formaldehyde and NH3 (Schutte et al. 1993a,b)
should significantly affect the amount of ammonium carbamate and
carbamic acid formed in interstellar ices.
1:1. Both
species desorb in the range 230-260 K. Many molecular clouds have
temperatures above 80 K and carbamic acid is therefore likely to
be present in the ISM, stored in its salt form, i.e. such as
ammonium carbamate [NH2COO-][NH4+] in the icy
mantle of the interstellar grains. This straightforward reaction
produces ammonium carbamate two of the most abundant species in
the ice mantle, NH3 and CO2. However, it is unlikely
that all the CO2 and NH3 present in interstellar grains will
be converted into ammonium carbamate and carbamic acid. In genuine
interstellar ices, a higher number of molecules are present in the
icy mantles, resulting in sophisticated and complex chemical
reaction network. The dilution in water ice should lower the
reaction rate. Competitive reaction, such as the thermal reaction
of formaldehyde and NH3 (Schutte et al. 1993a,b)
should significantly affect the amount of ammonium carbamate and
carbamic acid formed in interstellar ices.
Nevertheless, this thermal reaction between NH3 and CO2 in
interstellar conditions is relevant from an astrochemical point of
view because it provides the peptidic prototype HN-C=O linkage
without any energetic photons or particles. When the grain is
heated to above 230 K, during the protostellar disk formation
phase, ammonium carbamate decomposes directly into NH3 and
CO2, and the carbamic acid then sublimes and in turn
decomposes
![\begin{displaymath}[\textrm{NH}_4^+][\textrm{NH}_2\textrm{CO}_2^-] \rightarrow \textrm{CO}_2 + 2\times\textrm{NH}_3
\end{displaymath}](/articles/aa/full/2008/48/aa10536-08/img60.gif) |
(10) |
| (11) |
Our results also provide fresh insight that improves the study performed by Chen et al. (2007). In particular for the carbamic acid formation at 250 K. In this study, they irradiated at 15 K with UV/EUV photons, ice mixtures (12CO2:H2O, NH3:12CO2:H2O, NH3:13CO2:H2O) followed by warming to 250 K. Chen et al. (2007) proposed that carbamic acid could be formed from a thermal reaction between H2O and HNCO, photo-products which were observed after irradiation. On the basis of their spectra, HNCO is formed in such low quantity (absorbance of 0.02 or less) that it is unlikely to be the origin of carbamic acid that they observed at 250 K. As demonstrated previously Raunier et al. (2003), the thermal reaction between HNCO and H2O produces [H3O+][OCN-] at temperatures higher than 130 K. We propose that the products they observed at 250 K were produced mainly by thermal reaction between the residual NH3 and CO2.
A purely thermal reactivity occurs at low temperature, 80 K, for
NH3 and CO2, which are abundant in interstellar ices.
This implies that for molecular clouds with temperatures of around
100 K, ammonium carbamate and carbamic acid could be produced in
grains, and have a contribution to the ice absorption features at
3300 cm-1 (3 ![]() m) and 1500 cm-1 (6.6
m) and 1500 cm-1 (6.6 ![]() m). Once
desorbed at 250 K, carbamate and carbamic acid can also dissociate
easily into NH3 and CO2. This implies that ammonium
carbamate and carbamic acid must be incorporated into gas-grain
models as they store NH3 and CO2 in a stable salt form
until T>230 K at which they are a source of gas phase NH3and CO2, after H2O has desorbed. The amount of ammonium carbamate and carbamic acid should be affected by the composition of the ice due to competitive reactions.
m). Once
desorbed at 250 K, carbamate and carbamic acid can also dissociate
easily into NH3 and CO2. This implies that ammonium
carbamate and carbamic acid must be incorporated into gas-grain
models as they store NH3 and CO2 in a stable salt form
until T>230 K at which they are a source of gas phase NH3and CO2, after H2O has desorbed. The amount of ammonium carbamate and carbamic acid should be affected by the composition of the ice due to competitive reactions.
Acknowledgements
This work was supported by the PCMI (Physique et Chimie du Milieu Interstellaire) program and the CNES (Centre National d'Études spatiales).