W.-F. Thi1,2 - A. Bik1,3
1 - Sterrenkundig Instituut Anton Pannekoek,
University of Amsterdam, Kruislaan 403, 1098 SJ Amsterdam,
The Netherlands
2 -
ESA Research and Scientific Support Department, ESTEC, Keplerlaan 1, 2201 AZ Noordwijk, The Netherlands
3 -
European Southern Observatory, Karl-Schwarzschild-Strasse 2, 85748 Garching, Germany
Received 20 October 2004 / Accepted 18 March 2005
Abstract
We report on the observation of hot water vapor (steam) in
the inner AU of a young massive star located in the star-forming
region IRAS 08576-4334. The water lines are detected in a
medium resolution (
![]() )
K-band spectrum taken by the
infrared spectrometer ISAAC mounted on the VLT-ANTU.
The water vapor is at a mean temperature of
)
K-band spectrum taken by the
infrared spectrometer ISAAC mounted on the VLT-ANTU.
The water vapor is at a mean temperature of
![]() K, cooler
than the hot CO found in the same object, which is at
K, cooler
than the hot CO found in the same object, which is at ![]() 1660 K; and the column density is N(H2O
1660 K; and the column density is N(H2O
![]() cm-2. The profile of both H2O and CO lines is best reproduced by the emission from a Keplerian disk. To
interpret the data, we also investigate the formation of molecules
and especially CO and water vapor in the inner hot and dense part of
disks around young high mass stars using a pseudo time-dependent
gas-phase chemical model. Molecules are rapidly photodissociated
but this destruction is compensated for by an efficient formation
due to fast neutral-neutral reactions. The ability of CO molecules
to self-shield significantly enhances its abundance. Water molecules
are sufficiently abundant to be detectable. The observed H2O/CO ratio is reproduced by gas at 1600 K and by an enhanced UV field
over gas density ratio
cm-2. The profile of both H2O and CO lines is best reproduced by the emission from a Keplerian disk. To
interpret the data, we also investigate the formation of molecules
and especially CO and water vapor in the inner hot and dense part of
disks around young high mass stars using a pseudo time-dependent
gas-phase chemical model. Molecules are rapidly photodissociated
but this destruction is compensated for by an efficient formation
due to fast neutral-neutral reactions. The ability of CO molecules
to self-shield significantly enhances its abundance. Water molecules
are sufficiently abundant to be detectable. The observed H2O/CO ratio is reproduced by gas at 1600 K and by an enhanced UV field
over gas density ratio
![]() .
The
simulations support the presence of CO and H2O molecules in the
inner disks around young massive stars despite the strong UV radiation and show that the OH radical plays an essential role in
hot gas chemistry.
.
The
simulations support the presence of CO and H2O molecules in the
inner disks around young massive stars despite the strong UV radiation and show that the OH radical plays an essential role in
hot gas chemistry.
Key words: stars: formation - astrochemistry - circumstellar matter
Disks are found around many low-mass young stars and are natural byproducts of the star formation process (e.g., Waters & Waelkens 1998; Natta et al. 2000 for the Herbig Ae stars, and Mundy et al. 2000 for the T Tauri stars). Unfortunately, observations of disks around their higher counterparts remain inconclusive (Churchwell 2002). The presence or absence of a gaseous disk may help in understanding the detailed physics of high-mass star formation. One of the main difficulties in the formation of massive stars by direct molecular core collapse is that the radiation pressure of a luminous massive protostar will halt the accretion of matter, ultimately limiting the final mass of the star (Wolfire & Cassinelli 1987). Two major scenarii have been proposed to overcome this shortcoming. In the first one, massive stars can form by coagulation of two lower mass objects in a dense star cluster (Bonnell & Bate 2002). In the second scenario, a massive circumstellar disk will allow a significant amount of matter to accrete to the star (Yorke & Sonnhalter 2002; Jijina & Adams 1996). Evidence of the presence of a dense gaseous disk close to the star NGC 2024-IRS 2 as presented by Lenorzer et al. (2004). The main argument is that the infrared excess observed in NGC 2024-IRS 2 can be best explained by emission from a dense gaseous disk extending up to 0.6 AU.
Knowledge of the structure and composition of the inner disk is essential for our understanding of the accretion of matter onto the star. At the high densities and temperatures (100-2000 K) characteristic of disks at 0.1-5 AU around young stars, molecules are expected to be sufficiently excited to produce a rich ro-vibrational spectrum in the near- and mid-infrared. CO overtone emission, which is most likely emitted by a disk, has been detected and analyzed by Scoville et al. (1983) toward the Orion BN object and by Chandler et al. (1995) toward NGC 2024-IRS. Disk models have been successful in modeling CO bandhead emission in low-mass, as well as high-mass, young stellar objects (Najita et al. 1996; Chandler et al. 1995; Kraus et al. 2000). Cooler CO fundamental emission, which probes the planet-forming region of disks, has been detected in a large number of low-mass pre-main-sequence stars (Blake & Boogert 2004; Najita et al. 2003; Brittain et al. 2003).
Recent observations of CO overtone emission from young early type
stars further support the disk hypothesis (Bik & Thi 2004;
Blum et al. 2005). The presence of CO molecules raises the question
of their formation and/or survival in the harsh environment close to
massive stars. Molecules other than CO are also expected to exist in
the inner few AU of disks. Carbon, nitrogen, and oxygen are the most
abundant elements after Hydrogen and Helium. At high temperatures and
low pressures the dominant equilibrium species of these elements are CO, N2, and H2O; at intermediate temperatures, CH4, N2, and H2O; and at low temperatures and high pressures, CH4, NH3,
and H2O (Lewis & Prinn 1980). In all cases water vapor
is the second molecular reservoir of oxygen after CO in the gas phase
at a temperature between its dissociation temperature (![]() 2500 K)
and its condensation temperature (
2500 K)
and its condensation temperature (![]() 100-150 K). Basically, all
gas-phase oxygen locked into H2O does not participate to further
reactions (e.g., Kaufman & Neufeld 1996;
Elitzur 1979). Chemical models of
protoplanetary disks abound in the literature for low-mass stars
(e.g., Aikawa et al. 1999;
Markwick et al. 2002;
Semenov et al. 2004), high-mass stars (e.g.,
Nguyen et al. 2002) or debris-disks (e.g.,
Kamp et al. 2003), although most studies were limited
to the region located beyond a few AU. Nevertheless, several studies
focus on the thermal-chemical structure of the inner disks around
young low mass stars (Glassgold et al. 2004).
100-150 K). Basically, all
gas-phase oxygen locked into H2O does not participate to further
reactions (e.g., Kaufman & Neufeld 1996;
Elitzur 1979). Chemical models of
protoplanetary disks abound in the literature for low-mass stars
(e.g., Aikawa et al. 1999;
Markwick et al. 2002;
Semenov et al. 2004), high-mass stars (e.g.,
Nguyen et al. 2002) or debris-disks (e.g.,
Kamp et al. 2003), although most studies were limited
to the region located beyond a few AU. Nevertheless, several studies
focus on the thermal-chemical structure of the inner disks around
young low mass stars (Glassgold et al. 2004).
Fundamental ro-vibrational and pure-rotational water lines have been observed in the envelope of high-mass young stellar objects using the Short- and Long-Wavelength Spectrometer on board the Infrared Space Observatory (e.g., Boonman et al. 2003; Wright et al. 2000; Helmich et al. 1996). They find that freeze-out of water onto grains predominates in the cold part of the envelope, while the high abundance of water in the warm part can only be explained by sublimation of water molecules.
Water emission lines have also been observed in the sub-millimeter domain with the Submillimeter Wave Astronomy Satellite (SWAS), but the beam was too large to allow the study of small objects like the disks around high mass young stellar objects. Surprisingly, SWAS data show that gaseous H2O and O2 are not the dominant carriers of elemental oxygen in cold molecular clouds; the oxygen remains in atomic form, providing strong constraints on chemical models (Melnick et al. 2000; Bergin et al. 2000).
Highly excited water lines, which are absent in our atmosphere, are detected from the ground in the atmosphere of M dwarfs (see Allard et al. 2001, and references therein), Sunspots (Zobov et al. 2000), and in the disk around the low-mass young star SVS 13 (Carr et al. 2004). In the latter object, CO bandhead emission has also been detected.
In this paper, we model the hot water emission lines detected in the disk around the massive young star 08576nr292 (IRAS 08576-4334). The young stellar object (YSO) 08576nr292 is one of the infrared sources detected in a survey of H II regions by Kaper et al. (2005). We also investigate the condition for formation and/or survival of molecules (mainly CO, H2O) in the gaseous disk around a massive young star. The inner disk around a young massive star is probably too hot for dust grains to survive, such that no H2 formation on the grain surface can occur. Moreover, far ultraviolet photons are no longer absorbed by dust so that photodissociation occurs at high column densities. In addition, the disk temperature and density are high, making the chemistry different compared to other environments. For example, endothermic reactions or reactions with an activation energy, mostly the neutral-neutral reactions, and three-body reactions may dominate the chemical network over the classical ion-molecule reactions. The chemistry scheme is probably closer to that in shocks or protostellar winds than that of a cold quiescent molecular cloud.
The paper is organized as follows: we first present the observations and the fit to the K-band spectrum in Sect. 2. Then we explain the chemical models in Sect. 3. In Sect. 4, we present and discuss the results of the numerical runs and then finally conclude in Sect. 5.
Identified by Liseau et al. (1992) as IRS 34 in their survey of IRAS
sources in the Vela Molecular Cloud, IRAS 08576-4334 is a
high-mass star forming-region with total bolometric luminosity
![]() = 4.35. The distance to the cloud has been
estimated to be around 700 pc. The extended H II region
IRAS 08576-4334 includes a radio continuum source known as
G265.151+01.454 (Walsh et al. 1999). 08576nr292 is one
of the near-IR sources detected in the J- and K-band image of
IRAS 08576-4334 by Kaper et al. (2005) and discussed in
Bik et al. (2004). The properties of 08576nr292 are listed in
Table 1. After correction by 12 mag
of extinction, as derived by Bik et al. (2004), the J0-K0 color of
08576nr292 is small (0.4). Because the intrinsic color of hot
stars is
= 4.35. The distance to the cloud has been
estimated to be around 700 pc. The extended H II region
IRAS 08576-4334 includes a radio continuum source known as
G265.151+01.454 (Walsh et al. 1999). 08576nr292 is one
of the near-IR sources detected in the J- and K-band image of
IRAS 08576-4334 by Kaper et al. (2005) and discussed in
Bik et al. (2004). The properties of 08576nr292 are listed in
Table 1. After correction by 12 mag
of extinction, as derived by Bik et al. (2004), the J0-K0 color of
08576nr292 is small (0.4). Because the intrinsic color of hot
stars is ![]() -0.2, the near-IR excess from 08576nr292 is
(J-K)=0.6, suggesting no strong hot dust emission.
-0.2, the near-IR excess from 08576nr292 is
(J-K)=0.6, suggesting no strong hot dust emission.
Table 1: Properties of 08576nr292. The position and observed magnitude and color are taken from Kaper et al. (2005). The extinction estimate and derived unreddened magnitude and color are taken from Bik et al. (2004).
The spectrum of 08576nr292, identified in the line of sight
to IRAS 08576-4334, was observed with ISAAC at the Very
Large Telescope ANTU in 2002 at a resolving power of 10 000 (slit
width of 0.3
![]() ). The near-infrared data were reduced in a
standard way using a combination of the dedicated software package
Eclipse and IRAF. We made use of flatfield taken during the night, and
arc frames were taken by the European Southern Observatory (ESO)
staff during the day.
). The near-infrared data were reduced in a
standard way using a combination of the dedicated software package
Eclipse and IRAF. We made use of flatfield taken during the night, and
arc frames were taken by the European Southern Observatory (ESO)
staff during the day.
Standard stars of spectral type A observed at similar airmass were
used to correct for telluric OH emission and absorption. The
wavelength calibration was achieved by using arc lines in combination
with atmospheric OH lines. The details of the observations and
datareduction are presented in Bik et al. (2004). The chosen setting
covers the spectral range from 2.280 till 2.400 micron so that the
four first CO bandheads (
![]() ,
where v is the vibrational
quantum number) can be observed. At resolving power 10 000 (
,
where v is the vibrational
quantum number) can be observed. At resolving power 10 000 (
![]() km s-1) the profile of the lines is not resolved and
a few lines may even be blended.
km s-1) the profile of the lines is not resolved and
a few lines may even be blended.
The spectrum shows four prominent CO bandhead emissions, which are
modeled in Bik & Thi (2004). The fit to the first CO bandhead is
shown in the lower panel of Fig. 1. The upper panel
displays the spectrum shortward of
![]()
![]() m in detail. The CO bandhead is best fitted by gas at 1660 K located in a
Keplerian rotating disk extending from 0.2 till 3.6 AU and viewed with
an angle of
m in detail. The CO bandhead is best fitted by gas at 1660 K located in a
Keplerian rotating disk extending from 0.2 till 3.6 AU and viewed with
an angle of
![]() with respect to the disk rotation axis. The
column density of CO is relatively large (N(CO
with respect to the disk rotation axis. The
column density of CO is relatively large (N(CO
![]() cm-3).
cm-3).
Several emission features can be seen blueward of the first CO bandhead (i.e.
![]() m), which we attribute to water
vapor at T>1000 K. The emission features were checked against
atmospheric transmission spectra to ensure no artifact is present.
Using spectroscopic parameters drawn from the compilations HITEMP
(Rothman et al. 2003) and those of
Tennyson et al. (2001), most lines (often blended) in the
2.280-2.2935
m), which we attribute to water
vapor at T>1000 K. The emission features were checked against
atmospheric transmission spectra to ensure no artifact is present.
Using spectroscopic parameters drawn from the compilations HITEMP
(Rothman et al. 2003) and those of
Tennyson et al. (2001), most lines (often blended) in the
2.280-2.2935 ![]() m region can be assigned to hot water emission
lines. HITEMP (High-Temperature molecular spectroscopic
database) contains water vapor spectroscopic parameters of lines that
have greater strength than
m region can be assigned to hot water emission
lines. HITEMP (High-Temperature molecular spectroscopic
database) contains water vapor spectroscopic parameters of lines that
have greater strength than
![]() cm-1/(molecule cm-2) at 1000 K, while the energy of the levels in
Tennyson et al. (2001) were carefully assessed using
energy levels derived from sophisticated variational methods. The
HITEMP database contains 1816 water lines in total between 2.278 and 2.292
cm-1/(molecule cm-2) at 1000 K, while the energy of the levels in
Tennyson et al. (2001) were carefully assessed using
energy levels derived from sophisticated variational methods. The
HITEMP database contains 1816 water lines in total between 2.278 and 2.292 ![]() m, which are sufficiently strong at
m, which are sufficiently strong at
![]() K, but only 10 lines have strength greater
than 10-23 cm-1/(molecule cm-2), which is most likely
the threshold for visual detection in the observed spectrum. The
detected strong water lines and their spectroscopic characteristics
are summarized in Table 2. The wavelength of
three of the features (lines 8, 9, and 10) matches those of hot water
lines detected by Carr et al. (2004) in the inner disk
around the low mass young star SVS 13. The spectrum of
SVS 13 obtained by Carr et al. (2004) has a
higher resolution but covers a much smaller spectral range. It should
be noticed that all 1816 lines are included in the synthetic spectra
described in Sect. 2.4, and the position of the
lines is indicated in Figs. 2 and 4.
K, but only 10 lines have strength greater
than 10-23 cm-1/(molecule cm-2), which is most likely
the threshold for visual detection in the observed spectrum. The
detected strong water lines and their spectroscopic characteristics
are summarized in Table 2. The wavelength of
three of the features (lines 8, 9, and 10) matches those of hot water
lines detected by Carr et al. (2004) in the inner disk
around the low mass young star SVS 13. The spectrum of
SVS 13 obtained by Carr et al. (2004) has a
higher resolution but covers a much smaller spectral range. It should
be noticed that all 1816 lines are included in the synthetic spectra
described in Sect. 2.4, and the position of the
lines is indicated in Figs. 2 and 4.
All strong detected lines arise from the first vibrational
excited symmetric (100) and antisymmetric (001) stretch level of
ortho-H2O. It is known that there is a near coincidence of the
frequencies of those two modes. The rotational levels are high
(12<J<18), as expected since only high level lines are not present in
our atmosphere and can be readily detected with ground telescopes. At
the resolving power of 10 000 (or resolution
![]() km s-1), the lines are unresolved and few of them blended. The
observed lines arise from levels with energy from 5000-9000 K. The
detected (001)-(000) transitions have
km s-1), the lines are unresolved and few of them blended. The
observed lines arise from levels with energy from 5000-9000 K. The
detected (001)-(000) transitions have
![]() and
and
![]() and are therefore of a-type. The
(100)-(000) transition is of b-type (
and are therefore of a-type. The
(100)-(000) transition is of b-type (
![]() and
and
![]() )
(see Table 4).
)
(see Table 4).
Water lines are clearly seen in this object because they are not
blended. The profile of emission lines coming from the inner part (<a few AU) of a disk in Keplerian rotation can have ![]() up to
up to ![]() 200-250 km s-1 (or
200-250 km s-1 (or
![]() m) if
viewed edge-on. In comparison, water lines wavelength positions are
often separated by less than 65 km s-1 (or 0.0005
m) if
viewed edge-on. In comparison, water lines wavelength positions are
often separated by less than 65 km s-1 (or 0.0005 ![]() m),
therefore the rotational broadening of all lines produces a blend of
features that can be mistaken for a continuum.
m),
therefore the rotational broadening of all lines produces a blend of
features that can be mistaken for a continuum.
![\begin{figure}
\par\includegraphics[width=8.8cm,clip]{2219fig1.eps}
\end{figure}](/articles/aa/full/2005/29/aa2219-04/Timg40.gif) |
Figure 1:
The lower panel shows the best fit to the CO first bandhead toward
08576nr292. In the upper part the observed spectrum is
shown. The spectrum is continuum-subtracted and normalized to the
peak of the 2-0 bandhead. The best fit of the CO-bandhead is
plotted in the lower part. The parameters of the fit are
discussed in Bik & Thi (2004). Features shortward of 2.2935 |
| Open with DEXTER | |
Table 2:
Strongest detected water emission lines. Various spectroscopic characteristics of the detected lines are also provided.
The last column lists estimates of the critical density
![]() assuming
assuming
![]() K and
K and
![]() cm-2.
cm-2.
![\begin{figure}
\par\includegraphics[width=8.8cm,clip]{2219fig2.eps}
\end{figure}](/articles/aa/full/2005/29/aa2219-04/Timg47.gif) |
Figure 2:
Best fit to the hot water lines with the slab model.
The upper spectrum is a blowup of the spectrum showed in Fig. 1 between 2.278 and 2.292 |
| Open with DEXTER | |
In this section we apply two different models to fit the observed spectrum. The first model is a simplified version of the disk model applied to the CO bandheads of this object by Bik & Thi (2004), and in it we assume that the gas is originating from an isothermal slab of gas. The second model used to fit the data is similar to the model used to fit the CO bandheads. The gas is located in a Keplerian rotating, isothermal disk. We begin describing the ro-vibrational transitions of water vapor and the transition selection rules.
The structure of the water molecule is described in molecular physics
textbooks, e.g., Bernath (1995). As a triatomic
asymmetric (near-oblate) top molecule, water has three vibrational
modes and three rotational modes. The vibrational levels are
characterized by three vibrational quantum numbers v1 (symmetric
stretching mode ![]() ), v2 (bending mode
), v2 (bending mode ![]() ), and v3(asymmetric stretching mode
), and v3(asymmetric stretching mode ![]() ). The energy of the lowest
vibrational levels is given in Table 3.
The rotational levels are labeled using the standard asymmetric top
notation
JKa Kc, where J is the main rotational quantum
number, Ka the prolate quantum number (projection of J along
the a axis), and Kc the oblate quantum number (projection of Jalong the c axis). Notice that Ka+Kc=J or J+1. The rotational
statistical weight is (gJ=2J+1). The total degeneracy is
g=gI gJ.
). The energy of the lowest
vibrational levels is given in Table 3.
The rotational levels are labeled using the standard asymmetric top
notation
JKa Kc, where J is the main rotational quantum
number, Ka the prolate quantum number (projection of J along
the a axis), and Kc the oblate quantum number (projection of Jalong the c axis). Notice that Ka+Kc=J or J+1. The rotational
statistical weight is (gJ=2J+1). The total degeneracy is
g=gI gJ.
Like H2, H2O has two hydrogen atoms each with a nuclear spin 1/2, which combine to a singlet (para-H2O) and triplet (ortho-H2O). Transitions between para and ortho states are highly forbidden. Ortho states have a nuclear statistical weight gI of 3 while para states have gI equal to 1. The sum Ka+Kc+v3 is odd for ortho states and even for para states.
Table 3: Lower energy levels of vibration of water molecule in cm-1 with ground energy level at 0 cm-1. The zero-point energy is 4500 cm-1. To convert the energy scale into K, multiply by 1.438768.
The selection rules for asymmetric tops depend on the components
(![]() ,
,
![]() ,
,
![]() )
of the
permanent dipole moment vector, along the a, b, and c principal
molecular axes. The vibrational selection rules are
)
of the
permanent dipole moment vector, along the a, b, and c principal
molecular axes. The vibrational selection rules are
![]() for
the strong fundamental transitions and
for
the strong fundamental transitions and
![]() ,
3, 4, ... for
the weaker overtone transitions. The rotational selection rules can be
divided into three general cases given in
Table 4 in addition to
,
3, 4, ... for
the weaker overtone transitions. The rotational selection rules can be
divided into three general cases given in
Table 4 in addition to
![]() .
.
Table 4:
Selection rules for the ro-vibration transitions in addition to
![]() .
The transitions in brackets are weaker than the main transitions. For ro-vibrational transitions, the parity of
Ka+Kc+v3 has to be
conserved. Transitions of type a only occur for oblate molecules,
whereas transitions of type c are only found for prolate molecules.
Both oblate and prolate molecules can have b-type transitions.
.
The transitions in brackets are weaker than the main transitions. For ro-vibrational transitions, the parity of
Ka+Kc+v3 has to be
conserved. Transitions of type a only occur for oblate molecules,
whereas transitions of type c are only found for prolate molecules.
Both oblate and prolate molecules can have b-type transitions.
For the spectral modeling of the water lines we employ a method
similar to the fit of the CO bandhead emission, which is best modeled
by a Keplerian disk seen at low inclination with respect to the
rotation axis. First we simplify the model further and assume that
the water vapor lines arise from a isothermal slab of gas at
temperature
![]() with column density
with column density
![]() .
The remaining free parameter is the turbulent velocity width dv.
Values for dv range from 5 to 25 km s-1. One should keep in
mind that the profiles are unresolved and thus dv is an
ill-constrained parameter. The turbulent profile is a Gaussian
function. For dv>7 km s-1, the gas is highly turbulent. The
transition data are taken from the compilation HITEMP, and the level
population is assumed to be at local thermodynamical equilibrium
(LTE).
.
The remaining free parameter is the turbulent velocity width dv.
Values for dv range from 5 to 25 km s-1. One should keep in
mind that the profiles are unresolved and thus dv is an
ill-constrained parameter. The turbulent profile is a Gaussian
function. For dv>7 km s-1, the gas is highly turbulent. The
transition data are taken from the compilation HITEMP, and the level
population is assumed to be at local thermodynamical equilibrium
(LTE).
An order of magnitude check of this last assumption can be performed.
The collisional de-excitation coefficients for the transition
u
![]() l is given by
l is given by
![]() ,
where
,
where
![]() is the de-excitation cross section and
is the de-excitation cross section and
![]() cm s-1 is the mean relative
velocity of H2O molecules. In the absence of published
cross sections for the transitions considered here, we assume that
cm s-1 is the mean relative
velocity of H2O molecules. In the absence of published
cross sections for the transitions considered here, we assume that
![]() is close to the geometrical cross section
is close to the geometrical cross section
![]() cm2 of the molecule. The rates of
spontaneous emission can be estimated from the Einstein coefficients
cm2 of the molecule. The rates of
spontaneous emission can be estimated from the Einstein coefficients
![]() ,
which range from 10-5 s-1 to
,
which range from 10-5 s-1 to
![]() s-1 in the K-band region. The critical densities are
approximately
s-1 in the K-band region. The critical densities are
approximately
![]() cm-3. The number density in the
inner part of disks is higher than 1010 cm-3 from the fit
to the CO bandhead in LTE model. Therefore only the strongest lines
in the K-band are in non-LTE. More specifically, the lines detected
here have moderately strong Einstein coefficients
(10
-2-10-1 s-1) and by consequence relatively low
critical densities
cm-3. The number density in the
inner part of disks is higher than 1010 cm-3 from the fit
to the CO bandhead in LTE model. Therefore only the strongest lines
in the K-band are in non-LTE. More specifically, the lines detected
here have moderately strong Einstein coefficients
(10
-2-10-1 s-1) and by consequence relatively low
critical densities
![]() ,
which are reported in the last
column of Table 2.
,
which are reported in the last
column of Table 2.
The best model fit to the data was obtained by minimizing the ![]() using a Simplex (Press et al. 1992) and a genetic
algorithm method (Pikaia,
Charbonneau 1995). The results of the two
methods were cross-checked to ensure that no local minima were
reached. The best fit using a slab model gives H2O gas at
using a Simplex (Press et al. 1992) and a genetic
algorithm method (Pikaia,
Charbonneau 1995). The results of the two
methods were cross-checked to ensure that no local minima were
reached. The best fit using a slab model gives H2O gas at
![]() K and column density of (
K and column density of (
![]() cm-2. The best fit parameters are gathered in
Table 5. Although the errors on the gas
temperature and column density are dominated by systematics, we
estimated the uncertainties in the two parameters following the method
described in Bevington & Robinson (2003). The derived errors
represent here an increase of
cm-2. The best fit parameters are gathered in
Table 5. Although the errors on the gas
temperature and column density are dominated by systematics, we
estimated the uncertainties in the two parameters following the method
described in Bevington & Robinson (2003). The derived errors
represent here an increase of ![]() by 1 from its value at the
minimum. Noteworthy is that the statistical significance of the errors
is difficult to assess. The column density of water is 3 orders of
magnitude lower than CO, while the temperature is only slightly lower
for water than CO. The intrinsic turbulent width dv is an
ill-constrained parameter, although a value of 5 km s-1 has been
found to provide the best results. On the other hand, the column
density and gas temperature are well constrained.
by 1 from its value at the
minimum. Noteworthy is that the statistical significance of the errors
is difficult to assess. The column density of water is 3 orders of
magnitude lower than CO, while the temperature is only slightly lower
for water than CO. The intrinsic turbulent width dv is an
ill-constrained parameter, although a value of 5 km s-1 has been
found to provide the best results. On the other hand, the column
density and gas temperature are well constrained.
Table 5: Parameters derived from the fit by the slab and disk model. The parameters for the CO bandhead fitting are derived in Bik & Thi (2004). Accuracy in the temperature and column density estimates is limited by systematic instead of statistical errors (see text for a fuller discussion).
The second model includes broadening effects caused by a disk viewed
with an angle of 27
![]() ,
as found by fitting the CO bandhead. The
slab model spectrum is convolved with a profile which takes the
Doppler shifts caused by hot water located in a Keplerian disk into
account. The disk extends from
,
as found by fitting the CO bandhead. The
slab model spectrum is convolved with a profile which takes the
Doppler shifts caused by hot water located in a Keplerian disk into
account. The disk extends from
![]() till
till
![]() ,
which are two parameters of the model. The
inclination is assumed equal to the value obtained by fitting the
first CO bandhead (
,
which are two parameters of the model. The
inclination is assumed equal to the value obtained by fitting the
first CO bandhead (
![]() ). Line emission from a Keplerian
disk has a double-peak profile. The simple convolution procedure is
justified for H2O column densities in the range
10
18-1020 cm-2 where the optical depth is
). Line emission from a Keplerian
disk has a double-peak profile. The simple convolution procedure is
justified for H2O column densities in the range
10
18-1020 cm-2 where the optical depth is
![]() (see Fig. 3). The central star is assumed to have a mass
of 6
(see Fig. 3). The central star is assumed to have a mass
of 6 ![]() (Bik et al. 2004).
(Bik et al. 2004).
The dust sublimation radius, i.e. the radius where dust grains attain 1500 K, should provide an upper boundary to outer radius
![]() ,
because beyond this radius dust opacity will
dominate over the gas opacity and neither water nor CO lines can be
observed. A total column density of gas of
,
because beyond this radius dust opacity will
dominate over the gas opacity and neither water nor CO lines can be
observed. A total column density of gas of ![]() 1025 cm-2,
derived by multiplying the CO column density by 104, results in a
dust opacity of
1025 cm-2,
derived by multiplying the CO column density by 104, results in a
dust opacity of
![]() in the K-band using the extinction
law of Weingartner & Draine (2001), which assumes a standard
gas-to-dust mass ratio; and RV=3.1. A CO abundance of 104 is
reached for a large range of physical conditions (see
Sect. 3). However, gas located in the upper
atmosphere of the disk can be warmer than the mid-plane. The total
luminosity toward 08576nr292 is estimated to be around 103
in the K-band using the extinction
law of Weingartner & Draine (2001), which assumes a standard
gas-to-dust mass ratio; and RV=3.1. A CO abundance of 104 is
reached for a large range of physical conditions (see
Sect. 3). However, gas located in the upper
atmosphere of the disk can be warmer than the mid-plane. The total
luminosity toward 08576nr292 is estimated to be around 103 ![]() for a star of mass
for a star of mass ![]() 6.0
6.0 ![]() .
A crude estimate
of the dust sublimation radius is
.
A crude estimate
of the dust sublimation radius is
![]() AU
AU ![]() 1 AU, where a dust sublimation
temperature of 1500 K is assumed.
1 AU, where a dust sublimation
temperature of 1500 K is assumed.
The results of the fit by a disk model is displayed in
Fig. 4. The quality of the data and the resolution
do not permit an accurate constraint on parameters such as
![]() and
and
![]() .
The gas temperature and
column density are, however, better bracketed because these two
parameters are set by the relative strength between lines at LTE. The
best fit parameters are provided in Table 5
together with that derived from fitting the first CO bandhead.
.
The gas temperature and
column density are, however, better bracketed because these two
parameters are set by the relative strength between lines at LTE. The
best fit parameters are provided in Table 5
together with that derived from fitting the first CO bandhead.
![\begin{figure}
\par\includegraphics[angle=90,width=8.8cm,clip]{2219fig3.ps}
\end{figure}](/articles/aa/full/2005/29/aa2219-04/Timg81.gif) |
Figure 3:
Typical line optical depth for the hot water lines with dv=5 km s-1,
|
| Open with DEXTER | |
![\begin{figure}
\par\includegraphics[width=8cm,clip]{2219fig4.eps}
\end{figure}](/articles/aa/full/2005/29/aa2219-04/Timg82.gif) |
Figure 4: Best fit to the water lines toward 08576nr292 using the disk model. The upper curve is the observed normalized and continuum subtracted spectrum. The middle one represents the best fit, while the lower curve is the synthetic spectrum before degradation at the resolution of the observed spectrum. Notice the double-peak profile for lines which are not blended. |
| Open with DEXTER | |
The slab and disk models give similar values for the water gas
temperature and column density. This closeness can be ascribed to the
fact that the lines are optically thin. The intensity of optically
thin emission lines is not sensitive to the actual geometry of the
object. A mean value between the two models for the gas and column
density is adopted in this discussion. The resulting error on the gas
temperature is the scatter between the two temperatures and is much
smaller than in the formal error analysis (15 K instead of 510 K). The
water has a mean temperature of
![]() K and column density
of (
K and column density
of (
![]() cm-2. Both models give
relatively similar values for the temperature and column density.
This proximity can be ascribed to the low optical thickness of the
water lines.
cm-2. Both models give
relatively similar values for the temperature and column density.
This proximity can be ascribed to the low optical thickness of the
water lines.
The fits using either a slab or a disk model match the moderate
signal-to-noise ratio spectrum relatively well except that both fail
to reproduce the strength of the peak at 2.287 ![]() m (line 6)
relative to that of at 2.2875 (line 7). The upper level energy of both
lines are similar such that a non-LTE effect can be excluded (see
Table 2). Moreover the Einstein Acoefficient is slightly lower for line 6 than 7. Therefore line 7
should appear stronger in most situations. The most likely
explanation for this discrepancy is that the observed emission at 2.287
m (line 6)
relative to that of at 2.2875 (line 7). The upper level energy of both
lines are similar such that a non-LTE effect can be excluded (see
Table 2). Moreover the Einstein Acoefficient is slightly lower for line 6 than 7. Therefore line 7
should appear stronger in most situations. The most likely
explanation for this discrepancy is that the observed emission at 2.287 ![]() m is a combination of hot water and H2 3-2 S(1) emission.
Indeed, a strong H2 1-0 S(1) line has been detected in
08576nr292 at 2.218
m is a combination of hot water and H2 3-2 S(1) emission.
Indeed, a strong H2 1-0 S(1) line has been detected in
08576nr292 at 2.218 ![]() m (Bik et al. 2004). The origin of
the H2 lines is not well-determined and a large contribution from
the extended H II region is probable. Alternatively, errors and
uncertainties in the Einstein-A coefficients may exist and can be
sufficiently large to explain the bad fit around 2.287
m (Bik et al. 2004). The origin of
the H2 lines is not well-determined and a large contribution from
the extended H II region is probable. Alternatively, errors and
uncertainties in the Einstein-A coefficients may exist and can be
sufficiently large to explain the bad fit around 2.287 ![]() m.
m.
The fit by a slab requires a higher turbulent width (dv=25 km s-1) than the disk model (dv=5 km s-1). This difference stems from the difficulty of constraining dv with unresolved lines. The unlikely high value of dv for the slab model and the simultaneous goodness of the fit to the CO and water lines with the Keplerian disk model favor the latter interpretation of the geometry of the hot CO and water emitting region. Synthetic spectra obtained with a Keplerian disk and supersonic values for dv give unsatisfactory fits. Therefore we can rule out a turbulent Keplerian disk.
The water vapor column density of (
![]() cm-2 and the gas temperature
cm-2 and the gas temperature
![]() K
are close to the values found around the low-mass young star
SVS 13 by Carr et al. (2004), which is a priori
unexpected since SVS 13 and 08576nr292 differ in
mass. This similarity stems from that fact that the H2O emission
comes from a region very close to SVS 13.
Carr et al. (2004) find that the emitting area extents out
to 0.3 AU, while the water vapor is emitted from 2 to 4 AU in the disk
around 08576nr292. The hot gas is located at a shorter
distance from the low-mass star than from the massive star.
K
are close to the values found around the low-mass young star
SVS 13 by Carr et al. (2004), which is a priori
unexpected since SVS 13 and 08576nr292 differ in
mass. This similarity stems from that fact that the H2O emission
comes from a region very close to SVS 13.
Carr et al. (2004) find that the emitting area extents out
to 0.3 AU, while the water vapor is emitted from 2 to 4 AU in the disk
around 08576nr292. The hot gas is located at a shorter
distance from the low-mass star than from the massive star.
The water vapor temperature is about 100-200 K lower than that of the
hot CO, in accordance with the findings of
Carr et al. (2004) for SVS 13, although the hot
CO is much warmer around SVS 13. This lower temperature is
also consistent with a large inner and outer radius
(Table 5). Interestingly, the water temperature is
close to the maximum dust sublimation temperature of 1500 K, and the
outer radius
![]() is much larger than the dust
sublimation radius of 1 AU, though the effect of large dust opacity in
the observed spectrum is absent. In theory the low contrast between
the line and continuum should have prevented any possible detection of
the weak, optically thin water emission lines in the presence of dust.
One possible explanation is the lack of dust grains close to the star
at 1300-1500 K. The large luminosity of B5 stars can push the dust
grains well beyond the dust sublimation radius through radiation
pressure. Scaling the figures computed by
Saija et al. (2003) to the radius, mass, and effective
temperature of a B5 star, we obtain values for the ratios of radiation
pressure force over gravity
is much larger than the dust
sublimation radius of 1 AU, though the effect of large dust opacity in
the observed spectrum is absent. In theory the low contrast between
the line and continuum should have prevented any possible detection of
the weak, optically thin water emission lines in the presence of dust.
One possible explanation is the lack of dust grains close to the star
at 1300-1500 K. The large luminosity of B5 stars can push the dust
grains well beyond the dust sublimation radius through radiation
pressure. Scaling the figures computed by
Saija et al. (2003) to the radius, mass, and effective
temperature of a B5 star, we obtain values for the ratios of radiation
pressure force over gravity ![]() which are much larger than 10 for
all types of grain compositions. We have not taken the viscous drag
generated by the friction between the dust grains and the ambient gas
into account, which may be important in the dense inner disk. The
absence of hot dust is also compatible with the relatively small
near-infrared excess E(J-K). As a result of the separation between
the gas and dust, the hot gas at
which are much larger than 10 for
all types of grain compositions. We have not taken the viscous drag
generated by the friction between the dust grains and the ambient gas
into account, which may be important in the dense inner disk. The
absence of hot dust is also compatible with the relatively small
near-infrared excess E(J-K). As a result of the separation between
the gas and dust, the hot gas at
![]() K would be deprived
of elemental oxygen that comes from the sublimation of silicate
grains. Lower oxygen abundance in the gas phase may significantly
affect the chemistry, but we postpone discussion of the chemistry to
Sect. 4.5. Another possible explanation of the
low continuum optical depth resides in the difference in dust grain
lifetime. The maximum grain lifetime at a given temperature is
proportional to its radius. Lamy (1974) estimated
that the maximum lifetime of a 1
K would be deprived
of elemental oxygen that comes from the sublimation of silicate
grains. Lower oxygen abundance in the gas phase may significantly
affect the chemistry, but we postpone discussion of the chemistry to
Sect. 4.5. Another possible explanation of the
low continuum optical depth resides in the difference in dust grain
lifetime. The maximum grain lifetime at a given temperature is
proportional to its radius. Lamy (1974) estimated
that the maximum lifetime of a 1 ![]() m radius silicate grain at 1300 K is
m radius silicate grain at 1300 K is ![]() 0.6 year and ten times shorter for a 0.1
0.6 year and ten times shorter for a 0.1 ![]() m
radius grain. Therefore, we can envision that only the larger grains
in the dust size distribution can survive in the region where
T=1300-1500 K, which results in lower dust opacity.
Alternatively, if the thermal coupling between the gas and the dust is
inefficient, especially in the upper atmosphere of disks, the gas can
be hotter than the dust (Glassgold et al. 2004). In
addition, the disk's upper atmosphere can be relatively dust-free due to
dust settling. This latter possibility may be at work because a
disk model with subsonic turbulent velocity is favored. Finally, it
should be kept in mind that a combination of dust settling and size
segregation is possible.
m
radius grain. Therefore, we can envision that only the larger grains
in the dust size distribution can survive in the region where
T=1300-1500 K, which results in lower dust opacity.
Alternatively, if the thermal coupling between the gas and the dust is
inefficient, especially in the upper atmosphere of disks, the gas can
be hotter than the dust (Glassgold et al. 2004). In
addition, the disk's upper atmosphere can be relatively dust-free due to
dust settling. This latter possibility may be at work because a
disk model with subsonic turbulent velocity is favored. Finally, it
should be kept in mind that a combination of dust settling and size
segregation is possible.
We have demonstrated the presence of hot CO and H2O in the inner 4 AU of a Keplerian disk orbiting a B5 star. In the next section, a detailed chemical model is built in order to establish whether molecules can form and survive in the harsh environment around a hot star.
The presence of relatively large amounts of H2O and CO in the inner disk around young early type stars raises the question of their origin.
Early B stars are the main contributors to the mean interstellar
radiation field. A critical part of the UV spectrum is the
912-1000 Å range used for the computation of H2 and CO photodissociation rates. We modeled the chemistry that occurs in the
HI regions using a pseudo-time dependent chemical code, i.e. all
photons below 912 Å have been absorbed. The main parameters of the
models are the gas temperature
![]() and density
and density
![]() ,
the strength of the UV field and abundances of
the species at time t=0. The temperature
(
,
the strength of the UV field and abundances of
the species at time t=0. The temperature
(
![]() K) and density range
(
K) and density range
(
![]() cm-3) considered in our
modeling correspond to the values derived by
Lenorzer et al. (2004) when fitting the spectral energy
distribution of NGC 2024/IRS 2.
cm-3) considered in our
modeling correspond to the values derived by
Lenorzer et al. (2004) when fitting the spectral energy
distribution of NGC 2024/IRS 2.
The radiation field experienced by a parcel of gas is dominated by emission from the central star diluted by the distance to the star and
by self-shielding. Because of proximity to the central star, the
disk can be considered flat and the radiation flux received by the
disk surface at distance R from the star of radius R* with
effective temperature T* is:
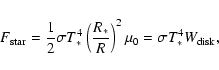 |
(1) |
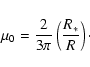 |
(2) |
The photodissociation rates in the UMIST database are multiplied by a
factor
![]() .
The spectrum of the mean interstellar UV field is
close to that of a B0V star, and it is assumed here that a unique
enhancement factor suffices to simulate the UV photodissociation. We
explore the effect of an enhanced UV field by factor
.
The spectrum of the mean interstellar UV field is
close to that of a B0V star, and it is assumed here that a unique
enhancement factor suffices to simulate the UV photodissociation. We
explore the effect of an enhanced UV field by factor
![]() because of the uncertainty in the
actual spectral type of 08576nr292 (Bik et al. 2004) and in
the inner and outer radius of the emitting area. In photodominated
chemistry, an important parameter is the ratio
because of the uncertainty in the
actual spectral type of 08576nr292 (Bik et al. 2004) and in
the inner and outer radius of the emitting area. In photodominated
chemistry, an important parameter is the ratio
![]() ,
which determines
the behavior of the chemistry. The energy balance of high-density
photodissociation regions is discussed by
Burton et al. (1990). The main cooling agents of gas at a
few thousand Kelvin are H2 ro-vibrational lines, followed by H2O lines. The heating is primarily provided by H2 vibrational heating.
Far UV photons will pump H2 molecules to a bound excited electronic
state (Lyman and Werner bands), from which they will fluoresce down to
a high vibrational level of the ground electronic state. Most of the
time (85%-90%) the excited H2 molecule will fluoresce back to a
low vibrational level while 10-15% of the time the molecule will
dissociate. At the high densities considered in this study, the
collision of excited H2 with other species contributes to the
heating of the gas and thermalization of the ro-vibrational levels.
,
which determines
the behavior of the chemistry. The energy balance of high-density
photodissociation regions is discussed by
Burton et al. (1990). The main cooling agents of gas at a
few thousand Kelvin are H2 ro-vibrational lines, followed by H2O lines. The heating is primarily provided by H2 vibrational heating.
Far UV photons will pump H2 molecules to a bound excited electronic
state (Lyman and Werner bands), from which they will fluoresce down to
a high vibrational level of the ground electronic state. Most of the
time (85%-90%) the excited H2 molecule will fluoresce back to a
low vibrational level while 10-15% of the time the molecule will
dissociate. At the high densities considered in this study, the
collision of excited H2 with other species contributes to the
heating of the gas and thermalization of the ro-vibrational levels.
We select molecular species composed of five atoms or less and
containing the elements H, C, O, N, Si, Mg, and Fe. The chemical
reactions are drawn from the compilation of UMIST rate99
(Le Teuff et al. 2000). In contrast to the interstellar
chemistry in cold environments, we expect that reactions with
activation barrier, endothermic, and three-body reactions dominate the
chemistry in inner hot disks. Therefore, besides the ion-molecules
reactions, two new types of reactions are taken into account: termolecular reactions, which are catalyzed bimolecular reactions
(i.e., 3-body reactions) and collider reactions, which are
collision-induced dissociations. To emphasize the likely importance of
3-body reactions, we provide the dissociation sequence of water by a
third body below,which is believed to be the initiation reaction in
the so-called Bradford sequence:
| (3) |
| (4) |
| (5) |
| (6) |
where M designates a third body (H, He, H2, ...). All together, the chemical network consists of 3299 reactions involving 301 species. Most species have negligible abundance (<10-30 w.r.t H). No deuterated species are included in our study.
Another major difference with previous studies of chemistry in disks around low- and intermediate-mass young stars (Aikawa et al. 1999; Markwick et al. 2002; Nguyen et al. 2002), which focus on the chemistry from a few AU (R>10 AU), is that the region modeled here is dust-free. The first consequence is the absence of the H2 formation reaction on grain surfaces. The second is that far ultraviolet photons are not absorbed by dust so that photodissociation is very effective.
Table 6: Initial abundances. The total oxygen budget is allowed to vary to explore the effect of different C/O elemental ratios. Water is assumed to be absent at t=0.
![\begin{figure}
\par\includegraphics[height=16cm,angle=90]{2219fig5.ps} %\end{figure}](/articles/aa/full/2005/29/aa2219-04/Timg112.gif) |
Figure 5:
Relative abundance of various species as a function of time and enhanced interstellar UV field
|
| Open with DEXTER | |
The model is static and isothermal, and it numerically solves a system
of differential equations that simulate the formation and destruction
of atomic and molecular species. A few solvers for systems of stiff
first order differential equations were used and their relative speed
was checked. The solvers are GEAR, VODE (Brown et al. 1989), and two
alternative algorithms described by Press et al. (1992).
All solvers give similar answers for a few test models, ensuring that
the results presented here are not numerical noise caused by a
specific solve. The adopted solver for the results presented in this
paper is VODE, the latest incarnation of the backward-differentiation
method popularized by GEAR. This solver gives the fastest results on
most of the models. We cover a parameter space in temperature
(1500-5000 K), density (
![]() cm-3), UV field, and CO self-shielding factor. The initial
abundance for the models is given in
Table 6, and the cosmic-ray flux is set
to the interstellar value. Models were run up to one year, which is
more than enough for the steady-state to be reached (see next
section).
cm-3), UV field, and CO self-shielding factor. The initial
abundance for the models is given in
Table 6, and the cosmic-ray flux is set
to the interstellar value. Models were run up to one year, which is
more than enough for the steady-state to be reached (see next
section).
After presenting the results of the chemical model calculations, we describe the formation routes of the main molecular species (H2, CO, and water) in a hot gas immersed in a strong UV field. The possibility that the molecules are formed in the outer part of the disk and transported into the inner few AU is discussed. Then we consider the effect of CO self-shielding against UV photodissociation and non-standard C/O abundance ratio on the molecular abundances. Finally the model predictions are compared to the observed abundances.
The abundance of H, H2, O, OH, H2O, C, C+, CO, and N are
shown in Fig. 5 for a gas temperature of 1600 K,
gas density of 1012 cm-3, and four values of
![]() (106, 107, 108, 109), neither CO nor
H2 self-shielding. First of all, it is clear that steady-state is
reached in less than a year. This rapidity is reassuring because the
matter located in the inner few AU of disks rapidly falls onto the
central star. As a comparison, the rotational period of the gas at
1 AU around a 6
(106, 107, 108, 109), neither CO nor
H2 self-shielding. First of all, it is clear that steady-state is
reached in less than a year. This rapidity is reassuring because the
matter located in the inner few AU of disks rapidly falls onto the
central star. As a comparison, the rotational period of the gas at
1 AU around a 6 ![]() star is
star is ![]() 150 days. In most cases,
steady-state is attained in a few days. The dominant species are the
atomic H, O, C, C+, N. Molecular hydrogen H2, CO, OH, and H2O are the most abundant radicals/molecules. Important abundance ratios
such as n(H)/n(H2), n(OH)/n(H2O), and n(CO)/n(H2O) are
displayed in Fig. 7 for different combinations of
150 days. In most cases,
steady-state is attained in a few days. The dominant species are the
atomic H, O, C, C+, N. Molecular hydrogen H2, CO, OH, and H2O are the most abundant radicals/molecules. Important abundance ratios
such as n(H)/n(H2), n(OH)/n(H2O), and n(CO)/n(H2O) are
displayed in Fig. 7 for different combinations of
![]() and
and
![]() (1600, 2000,
3000 and 4000 K). At
(1600, 2000,
3000 and 4000 K). At
![]() ,
the
chemistry is photo-regulated similar to a photodissociation region, and
all newly formed molecules are instantaneously destroyed. In the
thermal region (
,
the
chemistry is photo-regulated similar to a photodissociation region, and
all newly formed molecules are instantaneously destroyed. In the
thermal region (
![]() ), the
abundance of CO, OH, and H2O reduces with increasing gas temperature
because, once formed, those molecules react further to synthesize
other species. Therefore, the optimal temperature for molecular
abundance is around
), the
abundance of CO, OH, and H2O reduces with increasing gas temperature
because, once formed, those molecules react further to synthesize
other species. Therefore, the optimal temperature for molecular
abundance is around
![]() K.
K.
The chemical code monitors the main formation and destruction reactions at designated times. The formation routes for the main molecules are discussed below and a schematic is shown in Fig. 6. The chemical routes are different for dense and extremely dense gases. Below densities of around 109 cm-3 and at high temperature, the chemistry is dominated by two-body reactions with activation energy of the order of few thousand Kelvin, whereas at very high densities, three-body reactions dominate.
| |
Figure 6:
Schematic representation of the proposed chemical network. OH radicals play
a central role in this network. |
| Open with DEXTER | |
![\begin{figure}
\par\includegraphics[angle=90,width=16cm,clip]{2219fig7.ps}
\end{figure}](/articles/aa/full/2005/29/aa2219-04/Timg117.gif) |
Figure 7:
Relative abundance of various species as a function of
|
| Open with DEXTER | |
Molecular hydrogen is the starting molecule for this chemical network.
In the absence of dust grains, H2 is slowly synthesized in the gas
phase via three possible routes depending on the density and
temperature. At moderate densities, the reaction
| (7) |
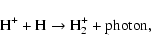 |
(8) |
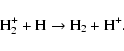 |
(9) |
| (10) |
CO molecules are readily formed for all values of the UV field
enhancement, when the density reaches a value of 106 cm-3 and
temperatures below 2500 K. The formation of CO is initiated by the
fast reaction at high temperature (activation energy of
![]() K)
K)
| (11) |
| (12) |
| (13) |
Above 3000 K, destruction of CO is enhanced because reactions with
high barrier are activated. Especially, CO is chemically dissociated
at gas temperature greater than 3000 K by the endothermic reaction:
| (14) |
At lower UV intensities, CO reacts with the OH+ to form HCO+. Here OH+ comes from the reaction between H3+ and atomic oxygen.
At extreme densities, CO is formed directly from the neutral-neutral
addition C+O, and the excess energy is carried away by a third body,
most likely a hydrogen atom. The main destruction reaction beside
photodissociation is the hydrogenation of CO leading to HCO. If the
abundance of H2 is not sufficiently high
| (15) |
| (16) |
| (17) |
| (18) |
The chemical reactions leading to water molecules in cold and hot
regions are different. In quiescent molecular clouds, the gas
temperature is below 230 K, and gas-phase chemistry gives typical
H2O abundances of a few ![]() 10-7 (e.g.,
Lee et al. 1996;
Le Teuff et al. 2000). Water is formed by a sequence
of ion-molecules reactions starting with
10-7 (e.g.,
Lee et al. 1996;
Le Teuff et al. 2000). Water is formed by a sequence
of ion-molecules reactions starting with
![]() or
or
![]() leading to OH+. Then rapid H-abstraction reactions with H2 produce H3O+ which dissociatively recombines (with electrons) to H2O. Another potential source of gas phase water molecules is the
evaporation of icy grains in regions where T>100 K. The reaction
scheme in cold regions has been proven to be too effective in
producing H2O when compared to observations of high-mass star
forming regions by Snell et al. (2000). At high
temperature (
leading to OH+. Then rapid H-abstraction reactions with H2 produce H3O+ which dissociatively recombines (with electrons) to H2O. Another potential source of gas phase water molecules is the
evaporation of icy grains in regions where T>100 K. The reaction
scheme in cold regions has been proven to be too effective in
producing H2O when compared to observations of high-mass star
forming regions by Snell et al. (2000). At high
temperature (
![]() K) and densities lower than 1015 cm-3, water molecules are predominately formed via the
radical-molecule reaction
K) and densities lower than 1015 cm-3, water molecules are predominately formed via the
radical-molecule reaction
| (19) |
| (20) |
Two main scenarii can explain the presence of molecules in the inner disks around young massive stars. The first model proposes that the molecules are formed at large distances from the central object and subsequently brought to its vicinity by accretion. We call this possibility the exogenous scenario. In the exogenous model, molecular species can also originate from evaporation of icy grain mantle. The second model states that molecules can be readily synthesized locally in the inner disks. This is the endogenous model. All models reach steady-state in at most one year and the effects of the initial molecular abundances are minimal.
At high column densities, CO molecules can self-shield from
photodissociation (e.g., van Dishoeck & Black 1988).
The nominal column of CO is 1015 cm-2 for a turbulent width
of 5 km s-1 but this value was derived for gas at low temperature
and may not be valid for gas above 1500 K, because highly vibrational
excited absorption levels were not taken into account in models of CO self-shielding. The nominal column of CO for self-shielding varies as 1/![]() .
For a very turbulent gas with
.
For a very turbulent gas with
![]() km s-1, the nominal column of CO is increased by a factor
km s-1, the nominal column of CO is increased by a factor ![]() 6.
Nevertheless, we ran models with increasing CO column density N(CO)
to account for probable increase in the value of the nominal CO for
self-shielding needed to simulate the condition in inner disks. The
other parameters are
6.
Nevertheless, we ran models with increasing CO column density N(CO)
to account for probable increase in the value of the nominal CO for
self-shielding needed to simulate the condition in inner disks. The
other parameters are
![]() K,
K,
![]() cm-3, and
cm-3, and
![]() .
Figure 10 shows that above the threshold CO column density of
.
Figure 10 shows that above the threshold CO column density of
![]() cm-2, CO molecules are
self-protected against photodissociation, and their abundance can
reach the cold quiescent molecular cloud value of
cm-2, CO molecules are
self-protected against photodissociation, and their abundance can
reach the cold quiescent molecular cloud value of ![]() 10-4.
Surprisingly, OH and H2O abundances seem to decrease when CO abundance rises steeply. The H2O/CO ratio drops dramatically from
few 10-4 to few 10-7. From
Fig. 8, it is clear that the effect of CO
self-shielding is most noticeable for
10-4.
Surprisingly, OH and H2O abundances seem to decrease when CO abundance rises steeply. The H2O/CO ratio drops dramatically from
few 10-4 to few 10-7. From
Fig. 8, it is clear that the effect of CO
self-shielding is most noticeable for
![]() (the photodominated regime).
The CO abundance reaches similar value to the thermal region, i.e.
where
(the photodominated regime).
The CO abundance reaches similar value to the thermal region, i.e.
where
![]() .
.
By pushing the dust grains beyond the dust sublimation radius, the radiation pressure will separate gas and dust. Segregation of the gas and dust results in an increase in elemental C/O ratio because the silicate grains, which contain 10-20% of the available oxygen, do not sublimate. The effect of an increase in the elemental C/O ratio above the solar value of 0.4 would be to lower the water and molecular oxygen abundance. In theory, the elemental abundances can be deduced from the study of the star's photospheric lines if it is not too embedded. Terzieva & Herbst (1998) discussed the different effects on the chemistry caused by variations in the C/O elemental abundance, but their study is limited to low temperature chemistry where elements can be depleted onto grains. Here we allow the oxygen's elemental abundance to vary from 0.1 to 1.0 times the nominal value listed in Table 6. The model results at steady-state are shown in Fig. 11. As expected, the relative abundance of CO, OH, and H2O diminish with decreasing oxygen elemental abundance. The rate of decrease is similar for the three species. Therefore, the relative abundances H2O/CO and H2O/OH are not significantly affected by changing the oxygen abundance. The simultaneous decrease in CO and H2O abundance with lower oxygen elemental abundance can be ascribed to the formation route of both molecules that are formed via the radial OH. This contrasts with the chemistry that occurs in quiescent molecular clouds where H2O is synthesized at a much lower rate than CO, which then results in a higher CO/H2O ratio as the oxygen elemental abundance decreases.
A large range of gas temperature (1600-4800 K) has been inferred from modeling CO bandhead emission toward four high-mass young stars (Bik & Thi 2004). The abundance of CO starts to decrease at temperature above 2500 K owing to collisional dissociation of CO and to the reaction of CO with OH to form CO2.
The observed H2O/CO ratio in 08576nr292 is
![]() .
If CO self-shielding is taken into account, the value for
.
If CO self-shielding is taken into account, the value for
![]() is not well-determined because the
theoretical curve for H2O/CO is relatively flat around few times 10-4 (upper left panel in Fig. 7). The CO abundance is 10
-7-10-4 and H2O abundance is 10
-11-10-8. Interestingly, when CO is allowed to
self-shield, we can infer
is not well-determined because the
theoretical curve for H2O/CO is relatively flat around few times 10-4 (upper left panel in Fig. 7). The CO abundance is 10
-7-10-4 and H2O abundance is 10
-11-10-8. Interestingly, when CO is allowed to
self-shield, we can infer
![]() for a gas at T=1600 K
better from the observed value of H2O/CO since the "degeneracy''
in the H2O/CO curve in the right panel of
Fig. 8 is lifted when CO self-shielding is
taken into account in the modeling.
for a gas at T=1600 K
better from the observed value of H2O/CO since the "degeneracy''
in the H2O/CO curve in the right panel of
Fig. 8 is lifted when CO self-shielding is
taken into account in the modeling.
![\begin{figure}
\par\resizebox{8.8cm}{!}{\includegraphics[angle=90]{2219fig8.eps}}
\end{figure}](/articles/aa/full/2005/29/aa2219-04/Timg143.gif) |
Figure 8:
Effect of CO self-shielding on abundance ratios. The left panel shows the addition of a strong self-shielding on the chemistry, while the right panel is the same as the upper right panel in Fig. 7. As expected the changes happen only on the H2O/CO and CO/H ratios. The observed value of H2O/CO ( |
| Open with DEXTER | |
![\begin{figure}
\par\resizebox{8.8cm}{!}{\includegraphics[angle=90]{2219fig9.ps}}
\end{figure}](/articles/aa/full/2005/29/aa2219-04/Timg144.gif) |
Figure 9:
H2O/CO versus H2O/OH diagram for various
|
| Open with DEXTER | |
![\begin{figure}
\par\resizebox{8.8cm}{!}{\includegraphics[angle=90]{2219fig10.ps}}
\end{figure}](/articles/aa/full/2005/29/aa2219-04/Timg145.gif) |
Figure 10:
Effect of the CO self-shielding on the relative abundances. The self-shielding factor
depends on the CO column density N(CO). The parameters of the
model are
|
| Open with DEXTER | |
![\begin{figure}
\par\resizebox{8.8cm}{!}{\includegraphics[angle=90]{2219fig11.ps}}
\end{figure}](/articles/aa/full/2005/29/aa2219-04/Timg146.gif) |
Figure 11:
Effect of the oxygen elemental abundance on the relative abundance of CO, OH, and H2O.
|
| Open with DEXTER | |
The absolute abundances are low in comparison to that in the envelope
of high-mass young stellar objects (Boonman et al. 2003).
From the CO abundance in the unshielded case, we infer a total
hydrogen column density of
![]() cm-2or surface density
cm-2or surface density
![]() g cm-2. When CO self-shielding effects are included in the modeling, CO/H reaches the
maximum relative abundance of 10-4 and the total hydrogen column
density amounts to
g cm-2. When CO self-shielding effects are included in the modeling, CO/H reaches the
maximum relative abundance of 10-4 and the total hydrogen column
density amounts to
![]() cm-2 or a surface
density of
cm-2 or a surface
density of
![]() g cm-2. We provide a large possible
range for the derived total hydrogen column density, because current CO self-shielding models may not be applicable to the situation
encountered in inner disks. However, it is noteworthy that the upper
range of column density (N(H)>1026 cm-2) would imply
large opacity in the near-infrared due to H-, which would prevent
the detection of optically thin H2O lines. The surface density is
comparable to the values adopted in disk models around T Tauri and
Herbig Ae stars, which is
g cm-2. We provide a large possible
range for the derived total hydrogen column density, because current CO self-shielding models may not be applicable to the situation
encountered in inner disks. However, it is noteworthy that the upper
range of column density (N(H)>1026 cm-2) would imply
large opacity in the near-infrared due to H-, which would prevent
the detection of optically thin H2O lines. The surface density is
comparable to the values adopted in disk models around T Tauri and
Herbig Ae stars, which is
![]() g cm-2at 1 AU.
g cm-2at 1 AU.
We summarize the chemical modeling in Fig. 9.
This figure shows the H2O/CO versus H2O/OH abundance ratio. The
figure provides a simple way to deduce physical parameters such as
![]() and
and
![]() from CO, OH,
and H2O abundance. Unfortunately, OH lines were not covered in the
K-band spectrum of 08576nr292.
from CO, OH,
and H2O abundance. Unfortunately, OH lines were not covered in the
K-band spectrum of 08576nr292.
Glassgold et al. (2004) studied the thermal-chemical structure of the inner disks around young low mass stars. Although in low-mass young stars X-rays drive the chemistry rather than the UV, they found that the disk atmosphere may contain a significant amount of warm ( T=500-2000 K) CO gas similar to our results. This similarity is not surprising since at the high temperatures and densities encountered in inner disks around low- and high-mass young stars, the chemistry is dominated by the same neutral-neutral reactions. At very high densities, the UV and X-ray photons provide the energy needed to maintain a high temperature. The photodissociation by X-ray or UV photons of the molecules are compensated for by rapid formation in dust-free regions. There, X-ray and UV photons are absorbed by gas, while in a dust-rich region UV radiation is rapidly attenuated by dust absorption while the X-ray can penetrate deeper to influence the ionization fraction (e.g., Semenov et al. 2003).
We have shown that hot-water vapor emission is present in the close vicinity (R<5 AU) of the high-mass young stellar object 08576nr292. The water vapor, as well as hot CO, are most likely located in a disk in Keplerian rotation.
The detection of optically thin CO and H2O lines at 1500-1600 K suggests that dust grains are absent in that temperature range in the disk, which may stem from the fact that dust grains are pushed further out by radiation pressure or that most dust has evaporated. The region probed by hot CO and H2O is limited to the inner few AU of the disk. Alternatively, the gas and dust may be thermally decoupled with the gas much warmer than the dust.
To probe cooler gas, which is located between 1 and ![]() 50 AU,
fundamental transitions of CO and lower J lines of H2O appear to
be more appropriate, although the line over continuum contrast would be
smaller owing to the warm dust emission. Nevertheless, hot lines allow
the gas-rich and probably dust-poor region to be investigated. Higher
resolution spectrometers (R=100 000 or
50 AU,
fundamental transitions of CO and lower J lines of H2O appear to
be more appropriate, although the line over continuum contrast would be
smaller owing to the warm dust emission. Nevertheless, hot lines allow
the gas-rich and probably dust-poor region to be investigated. Higher
resolution spectrometers (R=100 000 or
![]() km s-1),
such as CRIRES planned for the VLT, will resolve the
individual H2O line profiles. The combination of medium resolution
(R=10 000) and high spatial resolution achieved by AMBER will
help to determine the exact location of both the CO and H2O emissions.
km s-1),
such as CRIRES planned for the VLT, will resolve the
individual H2O line profiles. The combination of medium resolution
(R=10 000) and high spatial resolution achieved by AMBER will
help to determine the exact location of both the CO and H2O emissions.
Chemical models have been developed for the dense and hot inner disk
around young high-mass stars. The chemistry is dominated by
neutral-neutral reactions and steady-state is rapidly attained. H2 formation is hampered by the lack of dust grains and reaches relatively
low abundance. The radical OH is central to the chemical network
playing the same role as H3+ for cold chemistry. The molecular
abundances depend in general on the gas temperature and UV flux
over density ratio and show a large range of values.
Photodissociation affects more H2, OH, and H2O than CO because
the latter species can self-shield very efficiently. The observed
H2O/CO can be reproduced at the temperature of the gas (1600 K) and
![]() .
Observations of OH are
warranted in order to allow better constraints on the UV radiation field.
Future work on the modeling side includes the combination of the
chemical model presented here with a dynamic disk model around young
high-mass stars.
.
Observations of OH are
warranted in order to allow better constraints on the UV radiation field.
Future work on the modeling side includes the combination of the
chemical model presented here with a dynamic disk model around young
high-mass stars.
Detection of molecules such as CO and H2O at high temperature in the close vicinity of young massive young stellar objects opens the possibility of studying the physics and chemistry in the region where matter from the disk falls onto the star.
Acknowledgements
W.F.T. was supported by NWO grant 614.041.005 during his stay in Amsterdam. The authors thank the VLT staff for performing the observations in Service mode. Comments and suggestions from Rens Waters and Lex Kaper are much appreciated.