A&A 409, 159-161 (2003)
DOI: 10.1051/0004-6361:20031051
Infrared spectra of ZrC presolar grain analogues
Y. Kimura - C. Kaito
Department of Nanophysics in Frontier Project, Ritsumeikan University, Kusatsu-shi, Shiga 525-8577, Japan
Received 4 March 2003 / Accepted 16 June 2003
Abstract
The origins of spectral feature of stellar source are identified on the basis of laboratory studies. Although ZrC grains were identified as the crystalline presolar grains in meteorites, the spectral data of ZrC grains have not been measured in the laboratory. Therefore, ZrC grains with the same size as the presolar grains were produced in the laboratory. It has been found that the infrared spectrum of ZrC grains shows broad absorption features at 9.4 and 12.4 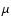 m.
m.
Key words: stars: AGB and post-AGB - stars: carbon - stars: circumstellar matter - ISM: general - infrared: ISM - infrared: stars
Presolar grains of carbide have been identified from the anomalous isotopic composition of carbon, nitrogen and silicon in SiC grains in the Murray carbonaceous meteorite (Bernatowicz et al. 1987). In addition to isotopic measurements, metallic (Ti, Mo, and Zr) carbides were identified in the 5-200 nm crystalline presolar grains by transmission electron microscopy (TEM) from graphitic spherules in Murchison meteorites (Bernatowicz et al. 1996). These metallic carbides were surrounded by well-graphitized carbon, i.e., many have a composite structure of a core of metallic carbides surrounded by a carbon mantle. Therefore in this paper, composite grains are denoted as core-mantle grains.
The origins of absorption peaks are identified on the basis of laboratory studies. Important factors governing the absorption features are size, shape and structure of the grain (Bohren & Huffman 1983). Therefore, the production of a presolar grain analogue is important for the identification of observed spectra.
One of the best methods of producing nanometer metallic carbide grains as presolar grains in the laboratory is the smoke experiment, which is a gas evaporation method (Uyeda 1987,1991). Using the gas evaporation method, solid grains are directly produced from the gas phase. We have produced metallic, oxide and sulfide grains by gas evaporation methods. The growth mechanism of these grains has been elucidated in a series of experiments (Kaito 1978,1981,1984,1985). However, since metallic carbide grains are refractory materials, their production is difficult. TiC core-carbon mantle grain formation was achieved in a previous study by introducing a noble gas evaporation method (Kimura & Kaito 2003). In the present experiment, we produced ZrC core-carbon mantle grains.
Although ZrC grains were identified in meteorites (Bernatowicz et al. 1996), the spectral data of ZrC grains have not been measured in the laboratory. Therefore, ZrC grains were produced in the present experiment. The characteristic spectrum is shown for ZrC.
![\begin{figure}
\par\includegraphics[width=8.8cm,clip]{Fig1.eps} \end{figure}](/articles/aa/full/2003/37/aa3675/Timg2.gif) |
Figure 1:
Experimental method of producing ZrC core-carbon mantle grains in smoke. Zirconium wire was set around the carbon electrode. |
| Open with DEXTER |
The work chamber used was a glass cylinder 17 cm in diameter and 30 cm in height covered with a stainless-steel plate and connected to a high-vacuum exhaust through a valve at its bottom. The present successful method of producing the ZrC grains is shown schematically in Fig. 1. Two carbon electrodes, P and Q, with a fine point, were prepared. The end of electrode Q was pushed with a spring. Zr wire was set around carbon electrode Q. After the chamber was evaporated to a pressure less than
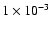 Pa, Ar gas was introduced at 13.3 kPa. By heating the carbon electrode in argon gas, the Zr wire was wet around the fine point of carbon electrode Q. As a result, since evaporation of carbon was suppressed, ZrC grains were obtained.
Pa, Ar gas was introduced at 13.3 kPa. By heating the carbon electrode in argon gas, the Zr wire was wet around the fine point of carbon electrode Q. As a result, since evaporation of carbon was suppressed, ZrC grains were obtained.
When the arc discharge is initiated in inert gas by this method, smoke rising from the evaporation source appears. The evaporated vapor is subsequently cooled and condensed in the gas atmosphere, i.e., solid grains are obtained directly from the gas cloud. Coalescent growth is regarded to be an important process in this method. Coalescence of the smoke particles has been explained in terms of two stages: a surface melting coalescence stage and a liquid-like coalescence stage among solid grains (Kaito 1978). These two stages depend mainly on grain size and temperature. The morphology and size of grains are controlled by the mass density and temperature of the smoke, which themselves depend on the atmospheric gas and gas pressure. By the same method, we succeeded in producing TiC core-carbon mantle grains of the same size as the core of presolar grains (Kimura & Kaito 2003).
The specimens were examined using a Hitachi H-7100R TEM. High-resolution transmission electron microscopy (HRTEM) images were obtained using a Hitachi H-9000NAR TEM. The infrared spectra of the samples embedded in KBr pellets were measured with a Fourier-transform infrared spectrometer (Horiba Inc. FT210). The wavelength resolution was 2 cm-1.
![\begin{figure}
\par\includegraphics[width=8.8cm,clip]{Fig2.eps} \end{figure}](/articles/aa/full/2003/37/aa3675/Timg4.gif) |
Figure 2:
Typical TEM image and corresponding ED pattern indicate the formation of ZrC grains.
|
| Open with DEXTER |
![\begin{figure}
\par\includegraphics[width=8.8cm,clip]{Fig3.eps} \end{figure}](/articles/aa/full/2003/37/aa3675/Timg5.gif) |
Figure 3:
Typical HRTEM image of a ZrC grain. The lattice distance and angles between closed lattice images indicate the existence of ZrC polycrystals.
|
| Open with DEXTER |
![\begin{figure}
\par\includegraphics[width=8.8cm,clip]{Fig4.eps} \end{figure}](/articles/aa/full/2003/37/aa3675/Timg6.gif) |
Figure 4:
Infrared spectra of ZrC grains. a) Spectrum for ZrC grains shown in Fig. 3. Broad absorption peak at 9.4 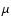 m is clearly observed. b) Spectrum of commercial Nilaco ZrC powder shown in Fig. 5. m is clearly observed. b) Spectrum of commercial Nilaco ZrC powder shown in Fig. 5.
|
| Open with DEXTER |
Figure 2 shows a typical TEM image and corresponding electron diffraction (ED) pattern of the collected specimen obtained by simultaneous evaporation of zirconium and carbon, as shown in Fig. 1. The ED pattern shows the formation of ZrC grains. A typical HRTEM image of a ZrC grain is shown in Fig. 3. The lattice spacing and angles between crossed lattices in the image indicate the existence of ZrC polycrystals, i.e., ZrC grains were composed of 5 nm crystallites. The external shape becomes spherical in spite of the NaCl-type structure, because the particles were compounds of crystallites and had many defects. Amorphous carbon grains with a size of 20 nm were also seen. Therefore, a carbon-rich condition is formed during ZrC growth. More than 50% of ZrC grains were surrounded by a carbon mantle. Zirconium grains are negligible. Figure 4a shows the infrared spectrum of the collected ZrC grains embedded in KBr pellets. The spectrum has a broad absorption feature at 9.4 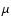 m. A very weak hump at the 12.4
m. A very weak hump at the 12.4 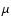 m absorption feature is also seen. Amorphous carbon grains were produced and their spectra were measured. The only absorption feature was a broad peak at 8.3
m absorption feature is also seen. Amorphous carbon grains were produced and their spectra were measured. The only absorption feature was a broad peak at 8.3 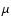 m; the peaks at 9.4 and 12.4
m; the peaks at 9.4 and 12.4 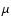 m did not appear. Therefore, the absorption at 9.4 and 12.4
m did not appear. Therefore, the absorption at 9.4 and 12.4 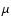 m was identified to be of ZrC origin. Since the ZrC presolar grains found in meteorites are nm-sized and coated with carbon, 9.4 and 12.4
m was identified to be of ZrC origin. Since the ZrC presolar grains found in meteorites are nm-sized and coated with carbon, 9.4 and 12.4 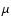 m features correspond to the ZrC presolar grains.
m features correspond to the ZrC presolar grains.
![\begin{figure}
\par\includegraphics[width=8.8cm,clip]{Fig5.eps} \end{figure}](/articles/aa/full/2003/37/aa3675/Timg7.gif) |
Figure 5:
Typical TEM image and ED pattern of commercial Nilaco ZrC powder.
|
| Open with DEXTER |
The infrared spectrum of commercial Nilaco ZrC powder was never observed within the limits of our measurements (3-25 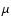 m), as shown in Fig. 4b. The size of the measured powder was 0.3-5
m), as shown in Fig. 4b. The size of the measured powder was 0.3-5 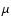 m which was separated by dispersion in ethanol. Typical Nilaco ZrC powder grains are shown in the TEM image in Fig. 5. Therefore, the absorption peak may be weakened by the increasing size of ZrC grains. This indicates that infrared absorption of the NaCl-type structure is weaker than that of any other structure, as discussed with respect to the mixture film of MgO and SiO (Suzuki et al. 2000).
m which was separated by dispersion in ethanol. Typical Nilaco ZrC powder grains are shown in the TEM image in Fig. 5. Therefore, the absorption peak may be weakened by the increasing size of ZrC grains. This indicates that infrared absorption of the NaCl-type structure is weaker than that of any other structure, as discussed with respect to the mixture film of MgO and SiO (Suzuki et al. 2000).
The TiC clusters with 27 to 125 atoms show a feature at a wavelength of 20.1 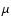 m (von Helden et al. 2000). On the other hand, a recent report on the TiC spectrum, in which infrared reflectance of TiC bulk material is shown, suggested that TiC are not responsible for the 20.1
m (von Helden et al. 2000). On the other hand, a recent report on the TiC spectrum, in which infrared reflectance of TiC bulk material is shown, suggested that TiC are not responsible for the 20.1 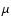 m feature(Henning & Mutschke 2001). Although TiC core (50 nm)-carbon mantle (2 nm) grains were thought to be at 9.5 and 12.5
m feature(Henning & Mutschke 2001). Although TiC core (50 nm)-carbon mantle (2 nm) grains were thought to be at 9.5 and 12.5 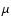 m, 21
m, 21 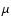 m was not included (Kimura & Kaito 2003). The 21
m was not included (Kimura & Kaito 2003). The 21 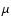 m feature of the gas phase TiC cluster by von Helden may be remarkable effect in cluster size (von Helden et al. 2000), as is discussed in a previous paper (Kimura & Kaito 2003). Therefore, 9.4 and 12.4
m feature of the gas phase TiC cluster by von Helden may be remarkable effect in cluster size (von Helden et al. 2000), as is discussed in a previous paper (Kimura & Kaito 2003). Therefore, 9.4 and 12.4 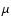 m features of ZrC grains may be due to small grain size. Size dependence of ZrC grains was very similar to the result of TiC grains. ZrC and TiC have the same crystal structure as NaCl against covalently bound SiC. Since the absorption of SiC is strong against as the continuum, the spectra can be easily observed. Consideration of the absorption intensity on bounding and crystal structure may become an important factor. The appearance of the 21
m features of ZrC grains may be due to small grain size. Size dependence of ZrC grains was very similar to the result of TiC grains. ZrC and TiC have the same crystal structure as NaCl against covalently bound SiC. Since the absorption of SiC is strong against as the continuum, the spectra can be easily observed. Consideration of the absorption intensity on bounding and crystal structure may become an important factor. The appearance of the 21 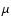 m feature in the TiC cluster may occur on ZrC for the very small clusters in gas phases. Since the absorption of ZrC is very weak compared to the continuum, the feature on ZrC may be unlikely to be detected by the observational spectrograph techniques in spite of its existence in meteorites.
m feature in the TiC cluster may occur on ZrC for the very small clusters in gas phases. Since the absorption of ZrC is very weak compared to the continuum, the feature on ZrC may be unlikely to be detected by the observational spectrograph techniques in spite of its existence in meteorites.
- Bernatowicz, T., Fraundorf, G., Ming, T., et al. 1987, Nature, 330, 728
In the text
NASA ADS
- Bernatowicz, T. J., Cowsik, R., Gibbons, P. C., et al. 1996, ApJ, 472, 760
In the text
NASA ADS
- Bohren, C. F., & Huffman, D. R. 1983, Absorption and Scattering of Right by Small Particles (Wiley Interscience)
In the text
- Henning, Th., & Mutschke, H. 2001, AcSpe A, 57, 815
In the text
- Kaito, C. 1978, JaJAP, 17, 601
In the text
- Kaito, C. 1981, JCrGr, 55, 273
In the text
- Kaito, C. 1984, JaJAP, 23, 525
In the text
- Kaito, C. 1985, JaJAP, 24, 261
In the text
- Kimura, Y., & Kaito, C. 2003, in press
In the text
- Kubo, R. 1962, J. Phys. Soc. Jpn., 17, 975
- Oshima, C., Aizawa, T., Wuttig, M., et al. 1987, Phys. Rev. B, 36, 7510
NASA ADS
- Suzuki, N., Kimura, S., Nakada, T., et al. 2000, M&PS, 35, 1269
In the text
NASA ADS
- Uyeda, R. 1987, in Morphology of Crystal, Part B, ed. I. Sunagawa, 369 (Tokyo Terra)
In the text
- Uyeda, R. 1991, Methods of preparation and technical applications. Prog. in Material Science 35, 1-96 (Pergamon Press)
In the text
- von Helden, G., Tielens, A. G. G. M., von Heijnsbergen, D., et al. 2000, Science, 288, 313
In the text
NASA ADS
Copyright ESO 2003
![\begin{figure}
\par\includegraphics[width=8.8cm,clip]{Fig1.eps} \end{figure}](/articles/aa/full/2003/37/aa3675/Timg2.gif)
![\begin{figure}
\par\includegraphics[width=8.8cm,clip]{Fig2.eps} \end{figure}](/articles/aa/full/2003/37/aa3675/Timg4.gif)
![\begin{figure}
\par\includegraphics[width=8.8cm,clip]{Fig3.eps} \end{figure}](/articles/aa/full/2003/37/aa3675/Timg5.gif)
![\begin{figure}
\par\includegraphics[width=8.8cm,clip]{Fig4.eps} \end{figure}](/articles/aa/full/2003/37/aa3675/Timg6.gif)
![\begin{figure}
\par\includegraphics[width=8.8cm,clip]{Fig5.eps} \end{figure}](/articles/aa/full/2003/37/aa3675/Timg7.gif)