Table 1:
Basic properties of the selected PAHs: ionization potential IP (eV)
and number of carbon atoms 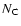 .
The two adopted types (1) and (2) correspond to two PAH symmetry groups (i.e.
geometric structure) (D6h coronenes and D2h acenes) that have a different IP-
.
The two adopted types (1) and (2) correspond to two PAH symmetry groups (i.e.
geometric structure) (D6h coronenes and D2h acenes) that have a different IP-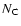 dependence. Type (2) PAHs are more easily ionized than type (1).
See also Fig. B.1. in Ruiterkamp et al. (2005).
We assume a generic electron affinity of 3 eV, and a sticking coefficient of unity.
dependence. Type (2) PAHs are more easily ionized than type (1).
See also Fig. B.1. in Ruiterkamp et al. (2005).
We assume a generic electron affinity of 3 eV, and a sticking coefficient of unity.
|
Source LaTeX |
All tables |
In the text

