A&A 403, 1003-1010 (2003)
DOI: 10.1051/0004-6361:20030364
A. M. S. Boonman 1 - E. F. van Dishoeck 1
Sterrewacht Leiden, PO Box 9513, 2300 RA Leiden, The Netherlands
Received 17 December 2002 / Accepted 25 February 2003
Abstract
We present infrared spectra of gas-phase H2O around 6 ![]() m toward
12 deeply embedded massive
protostars obtained with the Short Wavelength Spectrometer on board the
Infrared Space Observatory (ISO). The
m toward
12 deeply embedded massive
protostars obtained with the Short Wavelength Spectrometer on board the
Infrared Space Observatory (ISO). The ![]() ro-vibrational band has been
detected toward 7 of the sources and the excitation temperatures indicate
an origin in the warm gas at
ro-vibrational band has been
detected toward 7 of the sources and the excitation temperatures indicate
an origin in the warm gas at
![]() K.
Typical derived gas-phase H2O abundances are
K.
Typical derived gas-phase H2O abundances are
![]()
![]() ,
with the abundances
increasing with the temperature of the warm gas. The inferred gas/solid
ratios show a similar trend with temperature and suggest that
grain-mantle evaporation is important. The increasing gas/solid ratio
correlates with other indicators of
increased temperatures. If the higher temperatures are due to
a larger ratio of source luminosity to envelope mass,
this makes gas-phase H2O a good
evolutionary tracer. Comparison with
chemical models shows that three different chemical processes,
ice evaporation, high-T chemistry, and shocks, can reproduce the high
inferred gas-phase H2O abundances.
In a forthcoming paper each of these processes are investigated
in more detail in comparison with data from the Long Wavelength Spectrometer
on board ISO and the Submillimeter Wave Astronomy Satellite (SWAS).
Comparison with existing SWAS data indicates that a jump in the H2O abundance is present and that the observed
,
with the abundances
increasing with the temperature of the warm gas. The inferred gas/solid
ratios show a similar trend with temperature and suggest that
grain-mantle evaporation is important. The increasing gas/solid ratio
correlates with other indicators of
increased temperatures. If the higher temperatures are due to
a larger ratio of source luminosity to envelope mass,
this makes gas-phase H2O a good
evolutionary tracer. Comparison with
chemical models shows that three different chemical processes,
ice evaporation, high-T chemistry, and shocks, can reproduce the high
inferred gas-phase H2O abundances.
In a forthcoming paper each of these processes are investigated
in more detail in comparison with data from the Long Wavelength Spectrometer
on board ISO and the Submillimeter Wave Astronomy Satellite (SWAS).
Comparison with existing SWAS data indicates that a jump in the H2O abundance is present and that the observed ![]() ro-vibrational band traces primarily the warm inner envelope.
ro-vibrational band traces primarily the warm inner envelope.
Key words: ISM: abundances - ISM: molecules - molecular processes - stars: formation - stars: circumstellar matter - infrared: ISM
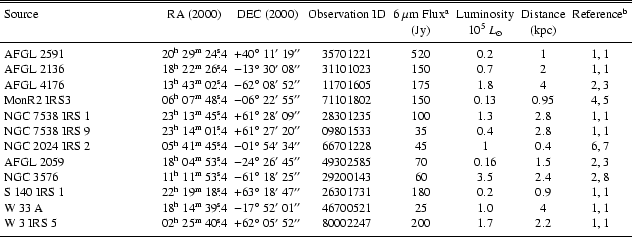 |
|
References: 1. Van der Tak et al. (2000b); 2. Lahuis & van Dishoeck (2000); 3. Henning et al. (1990); 4. Henning et al. (1992); 5. Giannakopoulou et al. (1997); 6. Thompson et al. (1981); 7. Anthony-Twarog (1982); 8. Persi et al. (1987). |
In recent years, many gas-phase H2O lines have been detected toward
star-forming regions. Prior to the Infrared Space Observatory (ISO), only a
few H2O and/or isotopic lines had been observed at submillimeter
wavelengths (e.g. Phillips et al. 1980;
Cernicharo et al. 1994;
Zmuidzinas et al. 1995; Gensheimer et al. 1996).
Observations of intermediate- to high-mass protostars with the
Short Wavelength Spectrometer (SWS) on board ISO have led to
the detection of
more than 30 different ro-vibrational H2O absorption lines around 6 ![]() m
(van Dishoeck & Helmich 1996;
Dartois et al. 1998; González-Alfonso et al. 1998).
The advantage of infrared absorption measurements is that many lines
can be observed at the same time in a pencil beam toward the infrared
source. This allows a much more accurate
determination of the gas-phase H2O abundance than from submillimeter
emission lines observed within different beams.
The disadvantage is the low spectral resolution of ISO of
m
(van Dishoeck & Helmich 1996;
Dartois et al. 1998; González-Alfonso et al. 1998).
The advantage of infrared absorption measurements is that many lines
can be observed at the same time in a pencil beam toward the infrared
source. This allows a much more accurate
determination of the gas-phase H2O abundance than from submillimeter
emission lines observed within different beams.
The disadvantage is the low spectral resolution of ISO of
![]() around 6
around 6 ![]() m,
so that absorption can only be detected toward sources with
intrinsic line widths of at least a few km s-1.
In addition, no kinematical information can
be derived from the data.
m,
so that absorption can only be detected toward sources with
intrinsic line widths of at least a few km s-1.
In addition, no kinematical information can
be derived from the data.
Abundances of up to ![]() 10-4 have been detected with ISO toward
intermediate- to high-mass star-forming regions
(van Dishoeck & Helmich 1996;
van Dishoeck 1998; Nisini et al.
1999; Ceccarelli et al. 1999).
These are in agreement with a variety of chemical models, including
high-T chemistry and shocks, which predict
enhanced H2O abundances
for
10-4 have been detected with ISO toward
intermediate- to high-mass star-forming regions
(van Dishoeck & Helmich 1996;
van Dishoeck 1998; Nisini et al.
1999; Ceccarelli et al. 1999).
These are in agreement with a variety of chemical models, including
high-T chemistry and shocks, which predict
enhanced H2O abundances
for ![]() -300 K where most of the oxygen is driven into H2O,
as well as grain-mantle evaporation predicting enhanced H2O abundances
for
-300 K where most of the oxygen is driven into H2O,
as well as grain-mantle evaporation predicting enhanced H2O abundances
for ![]() K
(Charnley 1997; Doty et al. 2002).
On the other hand, observations of the H2O
110-101 line
at 557 GHz obtained in a large (
K
(Charnley 1997; Doty et al. 2002).
On the other hand, observations of the H2O
110-101 line
at 557 GHz obtained in a large (![]() 4
4![]() )
beam with the
Submillimeter Wave Astronomy Satellite (SWAS)
show much lower gas-phase abundances of
)
beam with the
Submillimeter Wave Astronomy Satellite (SWAS)
show much lower gas-phase abundances of ![]() 10-9 to a few 10-8
in the cold (
10-9 to a few 10-8
in the cold (![]() K) gas
(Snell et al. 2000; Ashby et al. 2000).
K) gas
(Snell et al. 2000; Ashby et al. 2000).
So far, most studies of gas-phase H2O have focussed on a single or
a few source(s). In this paper, we present the analysis of the ![]() ro-vibrational
band of gas-phase H2O around
ro-vibrational
band of gas-phase H2O around ![]() 6
6 ![]() m toward 12 deeply embedded
massive protostars. The larger sample presented here
allows investigation of general evolutionary trends.
A detailed analysis of this
ro-vibrational H2O band in combination with
pure rotational lines observed with SWAS and the Long Wavelength Spectrometer
(LWS) on board ISO toward a sub-set of this
sample will be presented in a forthcoming paper
(Boonman et al. 2003b; hereafter Paper II).
m toward 12 deeply embedded
massive protostars. The larger sample presented here
allows investigation of general evolutionary trends.
A detailed analysis of this
ro-vibrational H2O band in combination with
pure rotational lines observed with SWAS and the Long Wavelength Spectrometer
(LWS) on board ISO toward a sub-set of this
sample will be presented in a forthcoming paper
(Boonman et al. 2003b; hereafter Paper II).
All sources in our sample have luminosities between ![]() 104-105
104-105
![]() and have been studied before in
other gas-phase molecules, both at infrared and submillimeter
wavelengths (e.g. Lahuis & van Dishoeck 2000;
Boonman et al. 2003a; van der Tak et al. 2003).
In addition, solid-state features of H2O, CO2 and 13CO2 toward the same sources have been analyzed (Keane et al. 2001b;
Gerakines et al. 1999; Boogert et al. 2000).
The reduction of the data is described in Sect. 2.
Section 3 describes the analysis of the spectra, using
pure absorption models, and radiative transfer effects are
investigated in Sect. 4. Finally, the
results are discussed in Sect. 5 and the conclusions
presented in Sect. 6.
and have been studied before in
other gas-phase molecules, both at infrared and submillimeter
wavelengths (e.g. Lahuis & van Dishoeck 2000;
Boonman et al. 2003a; van der Tak et al. 2003).
In addition, solid-state features of H2O, CO2 and 13CO2 toward the same sources have been analyzed (Keane et al. 2001b;
Gerakines et al. 1999; Boogert et al. 2000).
The reduction of the data is described in Sect. 2.
Section 3 describes the analysis of the spectra, using
pure absorption models, and radiative transfer effects are
investigated in Sect. 4. Finally, the
results are discussed in Sect. 5 and the conclusions
presented in Sect. 6.
The observations of the ![]() ro-vibrational band of gas-phase H2O around
6
ro-vibrational band of gas-phase H2O around
6 ![]() m have been performed with ISO-SWS
in the AOT6 grating mode, and are summarized in Table 1.
The spectra toward all sources have been reduced with the ISO-SWS Interactive Analysis System
SIA using the ISO Off-line Processing (OLP version 7) software
modules and calibration files.
Later versions of this Off-line Processing software (OLP versions 8 to 10)
have been checked and
no significant improvements are found. Unlike at other wavelengths, the
5-7
m have been performed with ISO-SWS
in the AOT6 grating mode, and are summarized in Table 1.
The spectra toward all sources have been reduced with the ISO-SWS Interactive Analysis System
SIA using the ISO Off-line Processing (OLP version 7) software
modules and calibration files.
Later versions of this Off-line Processing software (OLP versions 8 to 10)
have been checked and
no significant improvements are found. Unlike at other wavelengths, the
5-7 ![]() m region is relatively free from instrumental fringes, but since
defringing does improve the signal-to-noise ratio,
a cosine has been fitted to the data (Lahuis & van Dishoeck
2000). The depths of the H2O lines with and without
fringe removal have been compared to make sure that the
H2O lines are not partially removed by the defringing routine.
m region is relatively free from instrumental fringes, but since
defringing does improve the signal-to-noise ratio,
a cosine has been fitted to the data (Lahuis & van Dishoeck
2000). The depths of the H2O lines with and without
fringe removal have been compared to make sure that the
H2O lines are not partially removed by the defringing routine.
The spectra have been rebinned to an effective spectral resolution of
![]() m.
These spectra have been divided by a manual fit to the H2O ice
band resulting in the normalised spectra presented in
Fig. 1.
The 1
m.
These spectra have been divided by a manual fit to the H2O ice
band resulting in the normalised spectra presented in
Fig. 1.
The 1![]() noise level
in the final spectra is typically 1-3% of the continuum,
with the lowest noise level
corresponding to the sources with the highest continuum flux
(Table 1). The higher noise level between
noise level
in the final spectra is typically 1-3% of the continuum,
with the lowest noise level
corresponding to the sources with the highest continuum flux
(Table 1). The higher noise level between ![]() 5.7 and 6.2
5.7 and 6.2 ![]() m toward W 33 A is due to the presence of a strong H2O ice band
resulting in a sharp drop in the continuum to
m toward W 33 A is due to the presence of a strong H2O ice band
resulting in a sharp drop in the continuum to ![]() 10 Jy.
10 Jy.
![\begin{figure}
\par\includegraphics[width=15.7cm,clip]{ms3426f1.eps}
\end{figure}](/articles/aa/full/2003/21/aa3426/Timg24.gif) |
Figure 1:
Normalised ISO-SWS spectra of the |
The ![]() ro-vibrational band of gas-phase H2O has been detected
toward the sources AFGL 2591, AFGL 2136, AFGL 4176, MonR2 IRS3,
AFGL 2059 (M8E-IR), NGC 3576, and W 3 IRS5.
For AFGL 2591, AFGL 4176, and W 3 IRS5
the SWS AOT6 observations in parallel
with the AOT7 Fabry-Pérot scans have been used, since these have a
slightly better signal-to-noise ratio.
For sources for which multiple observations exist, the features are
very reproducable.
The spectra toward some of our sources
have been analysed previously by van Dishoeck & Helmich
(1996), Helmich et al. (1996),
and van Dishoeck (1998).
The reduced spectra presented here are however of a higher quality, since
both the instrument calibration and the reduction routines
have significantly improved for the 5-7
ro-vibrational band of gas-phase H2O has been detected
toward the sources AFGL 2591, AFGL 2136, AFGL 4176, MonR2 IRS3,
AFGL 2059 (M8E-IR), NGC 3576, and W 3 IRS5.
For AFGL 2591, AFGL 4176, and W 3 IRS5
the SWS AOT6 observations in parallel
with the AOT7 Fabry-Pérot scans have been used, since these have a
slightly better signal-to-noise ratio.
For sources for which multiple observations exist, the features are
very reproducable.
The spectra toward some of our sources
have been analysed previously by van Dishoeck & Helmich
(1996), Helmich et al. (1996),
and van Dishoeck (1998).
The reduced spectra presented here are however of a higher quality, since
both the instrument calibration and the reduction routines
have significantly improved for the 5-7 ![]() m wavelength region
compared to those used in 1996.
Also, the wavelength range shown here is somewhat larger
than in van Dishoeck & Helmich (1996), resulting in
the detection of more H2O absorption lines, especially between 6.6 and 7
m wavelength region
compared to those used in 1996.
Also, the wavelength range shown here is somewhat larger
than in van Dishoeck & Helmich (1996), resulting in
the detection of more H2O absorption lines, especially between 6.6 and 7 ![]() m.
The detection of the
m.
The detection of the ![]() ro-vibrational band of gas-phase H2O toward 7 sources and useful upper limits for the other massive
protostars will allow to search for evolutionary trends.
ro-vibrational band of gas-phase H2O toward 7 sources and useful upper limits for the other massive
protostars will allow to search for evolutionary trends.
The spectra in Fig. 1 are ordered according to
a decreasing temperature of the warm gas, with the warmest sources at the
top, using
![]() (C2H2) as a temperature indicator
(Lahuis & van Dishoeck 2000).
Generally, the strongest H2O absorption is found for the warmest sources.
For example,
the broad feature around 6.63
(C2H2) as a temperature indicator
(Lahuis & van Dishoeck 2000).
Generally, the strongest H2O absorption is found for the warmest sources.
For example,
the broad feature around 6.63 ![]() m appears to be strongest
for these sources. This feature is a blend of several gas-phase
lines with energy levels
m appears to be strongest
for these sources. This feature is a blend of several gas-phase
lines with energy levels ![]() 200 K, which can explain its absence
in the cooler sources. The warmer sources generally show more
lines for
200 K, which can explain its absence
in the cooler sources. The warmer sources generally show more
lines for
![]() m, which correspond to P-branch
lines from higher-J levels.
However, no gradual decrease of the absorption depth of the
m, which correspond to P-branch
lines from higher-J levels.
However, no gradual decrease of the absorption depth of the ![]() ro-vibrational band with decreasing temperature is seen.
ro-vibrational band with decreasing temperature is seen.
Most of the narrow absorption lines are in fact blends of two or more
lines arising from different energy levels, making it difficult to
find trends. The lines at ![]() 6.05 and
6.05 and ![]() 6.12
6.12 ![]() m, which include
the transitions from the ground-states of ortho- and para-H2O
respectively,
are clearly present in all sources where the
m, which include
the transitions from the ground-states of ortho- and para-H2O
respectively,
are clearly present in all sources where the ![]() ro-vibrational band
is detected. In NGC 7538 IRS1 and S 140 IRS1, where no detection is claimed,
there is a hint of the ground-state line of ortho-H2O.
ro-vibrational band
is detected. In NGC 7538 IRS1 and S 140 IRS1, where no detection is claimed,
there is a hint of the ground-state line of ortho-H2O.
The sources in which the ![]() ro-vibrational band of gas-phase
H2O is detected show all lines in absorption. Analysis of
the same band toward Orion-IRc2 shows the R-branch lines, with
ro-vibrational band of gas-phase
H2O is detected show all lines in absorption. Analysis of
the same band toward Orion-IRc2 shows the R-branch lines, with
![]() m, in absorption, whereas the P-branch
(
m, in absorption, whereas the P-branch
(
![]() m) is in emission (González-Alfonso et al.
1998). For this reason, we have not included Orion-IRc2 in the
analysis of the sample.
m) is in emission (González-Alfonso et al.
1998). For this reason, we have not included Orion-IRc2 in the
analysis of the sample.
The spectra have been modeled
using the method described in Lahuis & van Dishoeck (2000).
In this method, synthetic spectra are computed assuming that only absorption
takes place and that emission can be neglected. The effects of emission
filling in the absorption are discussed
in Sect. 4.
The source is assumed to be a homogeneous sphere with a single temperature
![]() and column density N, and
the molecular line data from the HITRAN 2000 database
(http://www.hitran.com) are used.
The models are more sensitive to the
H2O line width than those for other molecules.
Therefore, line widths of
and column density N, and
the molecular line data from the HITRAN 2000 database
(http://www.hitran.com) are used.
The models are more sensitive to the
H2O line width than those for other molecules.
Therefore, line widths of ![]() -10 km s-1
have been investigated.
The resulting synthetic spectra have been convolved to the nominal spectral
resolution of the ISO-SWS spectra
for comparison with the data.
-10 km s-1
have been investigated.
The resulting synthetic spectra have been convolved to the nominal spectral
resolution of the ISO-SWS spectra
for comparison with the data.
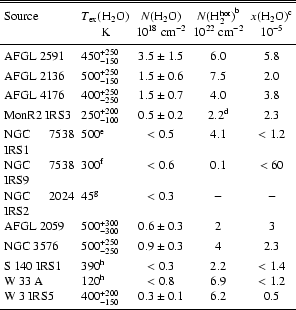 |
|
|
The best fit to the data has been
determined using the reduced
![]() -method.
Figure 2 shows an example of
-method.
Figure 2 shows an example of
![]() contours
for the source MonR2 IRS3 for different line widths.
This figure
illustrates that for low b-values (i.e.
contours
for the source MonR2 IRS3 for different line widths.
This figure
illustrates that for low b-values (i.e. ![]() km s-1) the
temperature and column density of gas-phase H2O are not well
constrained.
In the following analysis a line width of b=5 km s-1
is adopted for H2O.
This corresponds to the mean value of the 13CO v=1-0 absorption line
widths found by Mitchell et al. (1990).
For b=5 km s-1, the column density of gas-phase H2O is well
constrained, but the excitation temperature shows a
larger spread than for the CO2, HCN, and C2H2 absorption bands
using a similar analysis (Boonman et al. 2003a;
Lahuis & van Dishoeck 2000).
This is due to the absence of a Q-branch in the
km s-1) the
temperature and column density of gas-phase H2O are not well
constrained.
In the following analysis a line width of b=5 km s-1
is adopted for H2O.
This corresponds to the mean value of the 13CO v=1-0 absorption line
widths found by Mitchell et al. (1990).
For b=5 km s-1, the column density of gas-phase H2O is well
constrained, but the excitation temperature shows a
larger spread than for the CO2, HCN, and C2H2 absorption bands
using a similar analysis (Boonman et al. 2003a;
Lahuis & van Dishoeck 2000).
This is due to the absence of a Q-branch in the
![]() ro-vibrational band of H2O.
The best fitting model parameters for all sources are
listed in Table 2 and
Fig. 3 shows some good fitting H2O model spectra
for the source MonR2 IRS3.
ro-vibrational band of H2O.
The best fitting model parameters for all sources are
listed in Table 2 and
Fig. 3 shows some good fitting H2O model spectra
for the source MonR2 IRS3.
The results show that the detected H2O gas is warm
(
![]() K), but that there is not a clear correlation
with
K), but that there is not a clear correlation
with
![]() (C2H2)
(Fig. 4). This is probably caused by the large
error bars on the H2O excitation temperature.
Van der Tak (2000)
argues that the dust
opacity at 6
(C2H2)
(Fig. 4). This is probably caused by the large
error bars on the H2O excitation temperature.
Van der Tak (2000)
argues that the dust
opacity at 6 ![]() m is comparable to that at 13
m is comparable to that at 13 ![]() m, near the C2H2 absorption band. This would imply
that the lack of a clear correlation between
m, near the C2H2 absorption band. This would imply
that the lack of a clear correlation between
![]() (C2H2) and
(C2H2) and
![]() (H2O) is likely not
caused by hot gas-phase H2O hidden behind an opaque dust layer.
Plotting
(H2O) is likely not
caused by hot gas-phase H2O hidden behind an opaque dust layer.
Plotting
![]() (H2O) versus
(H2O) versus
![]() (CO)
of the warm gas from Mitchell et al. (1990)
shows the same lack of a clear correlation.
Figure 9 of Lahuis & van Dishoeck (2000) shows that overall
there is a good correlation between the CO and C2H2 temperatures.
In general,
(CO)
of the warm gas from Mitchell et al. (1990)
shows the same lack of a clear correlation.
Figure 9 of Lahuis & van Dishoeck (2000) shows that overall
there is a good correlation between the CO and C2H2 temperatures.
In general,
![]() (C2H2) is preferred as a tracer of
the inner warm envelope because it suffers less from confusion by
outflow material, and because the absorption of this molecule is enhanced in
the inner region.
(C2H2) is preferred as a tracer of
the inner warm envelope because it suffers less from confusion by
outflow material, and because the absorption of this molecule is enhanced in
the inner region.
![\begin{figure}
\par\includegraphics[width=8.4cm,clip]{ms3426f3.eps}
\end{figure}](/articles/aa/full/2003/21/aa3426/Timg45.gif) |
Figure 3:
Example of some good fitting models
for the observed |
The derived H2O column densities have been
converted into abundances
with respect to the warm H2 gas, since the spectra
show primarily warm H2O gas at
![]() K.
The H2 column densities in the warm gas have been
derived from infrared observations
of 13CO (e.g. Mitchell et al. 1990).
A 12CO/13CO ratio of 60 and a 12CO/H2 ratio of
K.
The H2 column densities in the warm gas have been
derived from infrared observations
of 13CO (e.g. Mitchell et al. 1990).
A 12CO/13CO ratio of 60 and a 12CO/H2 ratio of
![]() have been assumed (Lahuis & van Dishoeck 2000;
Lacy et al. 1994).
Typical derived gas-phase H2O abundances are
have been assumed (Lahuis & van Dishoeck 2000;
Lacy et al. 1994).
Typical derived gas-phase H2O abundances are
![]()
![]() ,
increasing with temperature (Fig. 5;
Table 2).
The derived H2O abundances for AFGL 2591, AFGL 2136, AFGL 4176, and
NGC 7538 IRS9 agree well with those previously derived by van Dishoeck
& Helmich (1996), but the derived H2O excitation
temperatures for these sources seem somewhat higher than their
adopted value of
,
increasing with temperature (Fig. 5;
Table 2).
The derived H2O abundances for AFGL 2591, AFGL 2136, AFGL 4176, and
NGC 7538 IRS9 agree well with those previously derived by van Dishoeck
& Helmich (1996), but the derived H2O excitation
temperatures for these sources seem somewhat higher than their
adopted value of
![]() K (Table 2).
K (Table 2).
The H2O ice abundances with respect to cold H2 gas are shown in
Fig. 5 for comparison and are seen to
decrease with temperature.
Using
![]() (H2O) instead of
(H2O) instead of
![]() (C2H2) in Fig. 5 shows the same
trends, although somewhat steeper.
This suggests that grain mantle evaporation plays an important role in
the production of gas-phase H2O.
(C2H2) in Fig. 5 shows the same
trends, although somewhat steeper.
This suggests that grain mantle evaporation plays an important role in
the production of gas-phase H2O.
![\begin{figure}
\par\includegraphics[width=6.3cm,clip]{ms3426f4.eps}
\end{figure}](/articles/aa/full/2003/21/aa3426/Timg48.gif) |
Figure 4:
Correlation between the H2O excitation temperature
and that of C2H2,
a good tracer of the warm gas (Lahuis & van
Dishoeck 2000). Only those sources are shown for which both
excitation temperatures are determined, except for
MonR2 IRS3 for which
|
The gas-phase
abundance in the warmest source is still a factor of ![]() 3.5 lower than
the ice abundance in the coldest source.
If the gas-phase H2O results from evaporation of ice mantles, they
are expected to be the same. The difference may have several reasons.
First, the adopted b-value may be too large. Adopting a lower b-value
for which the column density is still well-constrained
(b>2 km s-1), may increase the
inferred abundances by factors of
3.5 lower than
the ice abundance in the coldest source.
If the gas-phase H2O results from evaporation of ice mantles, they
are expected to be the same. The difference may have several reasons.
First, the adopted b-value may be too large. Adopting a lower b-value
for which the column density is still well-constrained
(b>2 km s-1), may increase the
inferred abundances by factors of ![]() 1.5. Second, part of the H2O absorption may originate in cold H2O gas.
Using the total H2 column density instead of the warm component only,
results in a factor of
1.5. Second, part of the H2O absorption may originate in cold H2O gas.
Using the total H2 column density instead of the warm component only,
results in a factor of ![]() 2.3 lower H2O abundances in the gas-phase compared
to the ices.
Fitting a two-temperature
component model to the data,
using the 13CO temperatures for the
warm and cold gas, shows that column densities up to
2.3 lower H2O abundances in the gas-phase compared
to the ices.
Fitting a two-temperature
component model to the data,
using the 13CO temperatures for the
warm and cold gas, shows that column densities up to ![]() 1018 cm-2 could be present in cold H2O gas at
1018 cm-2 could be present in cold H2O gas at ![]() K.
Such large amounts of cold gas should
have been picked up with the SWAS satellite, which is not the case
(see Paper II).
Therefore, it is not likely that the cold H2O gas below
K.
Such large amounts of cold gas should
have been picked up with the SWAS satellite, which is not the case
(see Paper II).
Therefore, it is not likely that the cold H2O gas below ![]() 100 K
contributes significantly to the observed absorption spectra.
A third possibility is that H2O may be destroyed
in the gas-phase after evaporation from the grains.
Current models of gas-phase chemistry predict production of
gas-phase H2O in warm regions rather than destruction (e.g. Charnley
1997). On the other hand, processes like X-ray dissociation
may be able to
destroy some gas-phase H2O in the warmest regions close to the
central radiation source, but this is not likely to be a significant fraction
(see Boonman et al. 2003a).
100 K
contributes significantly to the observed absorption spectra.
A third possibility is that H2O may be destroyed
in the gas-phase after evaporation from the grains.
Current models of gas-phase chemistry predict production of
gas-phase H2O in warm regions rather than destruction (e.g. Charnley
1997). On the other hand, processes like X-ray dissociation
may be able to
destroy some gas-phase H2O in the warmest regions close to the
central radiation source, but this is not likely to be a significant fraction
(see Boonman et al. 2003a).
Finally, both the assumption of pure absorption and of
a homogeneous source with a constant temperature
and column density may underestimate the H2O abundance in the warm gas.
The effect of emission filling in the absorption is discussed in
Sect 4.
In Paper II
it will be shown that combining the inferred temperature and density
gradients from van der Tak et al. (2000b) with a detailed
chemical model, results in H2O abundances of ![]() 10-4
in the warm gas and can successfully explain the observed
ro-vibrational spectrum of gas-phase H2O. Therefore, the difference
between the gas-phase H2O abundance in the warmest source
and the ice abundance in the coldest source is probably due to
both the assumption of pure absorption and of a homogeneous source with a
constant excitation temperature and column density in the current models.
10-4
in the warm gas and can successfully explain the observed
ro-vibrational spectrum of gas-phase H2O. Therefore, the difference
between the gas-phase H2O abundance in the warmest source
and the ice abundance in the coldest source is probably due to
both the assumption of pure absorption and of a homogeneous source with a
constant excitation temperature and column density in the current models.
![\begin{figure}
\par\includegraphics[width=8.4cm,clip]{ms3426f5.eps}
\end{figure}](/articles/aa/full/2003/21/aa3426/Timg52.gif) |
Figure 5:
The H2O gas-phase abundances from
Table 2 (left panel)
and the H2O ice abundances from Gibb & Whittet (2002)
and Gerakines et al. (1999) (right panel) versus
|
The derived gas-phase H2O abundances from Table 2
can be combined with the corresponding ice abundances from
Gibb & Whittet (2002) and Gerakines et al. (1999)
to determine the gas/solid ratios. Figure 6
shows that these ratios increase with temperature.
A similar trend is seen for CO2 (Boonman et al. 2003a).
Using
![]() (H2O) instead of
(H2O) instead of
![]() (C2H2) in Fig. 6 shows a similar,
but somewhat steeper trend.
(C2H2) in Fig. 6 shows a similar,
but somewhat steeper trend.
The low gas/solid ratios generally correspond to those sources
for which the ![]() ro-vibrational band of gas-phase H2O is not detected.
This indicates that for those sources either most of the H2O is still on the grains or that the line widths for these
sources are smaller than the adopted b=5 km s-1. A smaller line width
can increase the inferred gas/solid ratios for the colder sources
by a factor of a few, but will not change the general increase of the
gas/solid ratio with temperature.
The observed trend in the gas/solid ratio
emphasizes the result from Fig. 5
that grain mantle evaporation plays a role.
ro-vibrational band of gas-phase H2O is not detected.
This indicates that for those sources either most of the H2O is still on the grains or that the line widths for these
sources are smaller than the adopted b=5 km s-1. A smaller line width
can increase the inferred gas/solid ratios for the colder sources
by a factor of a few, but will not change the general increase of the
gas/solid ratio with temperature.
The observed trend in the gas/solid ratio
emphasizes the result from Fig. 5
that grain mantle evaporation plays a role.
In the previous sections it is assumed that only absorption takes place,
but especially in the warm gas, emission may fill in the absorption.
For example,
toward Orion-IRc2 part of the H2O ![]() band appears in
emission (González-Alfonso et al. 1998).
In order to investigate this effect, an excitation model
has been set-up using rotational energy levels up to
band appears in
emission (González-Alfonso et al. 1998).
In order to investigate this effect, an excitation model
has been set-up using rotational energy levels up to ![]() cm-1 in
the ground state and
cm-1 in
the ground state and
![]() cm-1 in
the first vibrationally excited state.
The level populations
are calculated adopting a Boltzmann distribution using
cm-1 in
the first vibrationally excited state.
The level populations
are calculated adopting a Boltzmann distribution using
![]() from Table 2.
As central radiation source, a blackbody
is adopted for each object
at a temperature
from Table 2.
As central radiation source, a blackbody
is adopted for each object
at a temperature ![]() 100 K higher than the
inferred H2O excitation temperature, to maximize the effect of
emission filling in the absorption lines. If the blackbody temperature is
taken equal to
100 K higher than the
inferred H2O excitation temperature, to maximize the effect of
emission filling in the absorption lines. If the blackbody temperature is
taken equal to
![]() (H2O) the P-branch lines appear in
emission, while the R-branch lines are still in absorption
similar to what is
observed toward Orion-IRc2 (González-Alfonso et al. 1998).
However,
our sources do not show such a phenomenon. This
is probably due to their larger distances compared
to Orion allowing a much smaller part of the beam being
filled with emission. This emission will be more beam-diluted and thus
more difficult to detect.
(H2O) the P-branch lines appear in
emission, while the R-branch lines are still in absorption
similar to what is
observed toward Orion-IRc2 (González-Alfonso et al. 1998).
However,
our sources do not show such a phenomenon. This
is probably due to their larger distances compared
to Orion allowing a much smaller part of the beam being
filled with emission. This emission will be more beam-diluted and thus
more difficult to detect.
Adopting a homogeneous source with a constant temperature and density
as in Sect. 3 for
b=5 km s-1, shows that emission starts filling in the absorption
significantly for
![]() K.
The resulting column densities are a factor
of
K.
The resulting column densities are a factor
of ![]() 3-6 higher than in the case of pure absorption.
3-6 higher than in the case of pure absorption.
Van der Tak et al. (2000b) and Mueller et al. (2002)
have shown that temperature
and density gradients are present in the envelopes of the massive protostars
studied here. Adopting the physical structure
from van der Tak et al. (2000b) and assuming a Boltzmann
distribution in each shell with
![]() equal to the dust
temperature in that shell increases the inferred column density
by similar factors as in the case of a constant temperature and density.
equal to the dust
temperature in that shell increases the inferred column density
by similar factors as in the case of a constant temperature and density.
These factors can easily account for the difference between the H2O
gas-phase abundances for the warmest sources and the H2O ice abundances
in the coldest sources.
Since the inclusion of emission enhances the abundances in particular for
the warmest sources, this effect
also preserves the inferred trends with
temperature for the gas-phase H2O abundances and gas/solid ratios
(see Sect. 3).
It should be noted that it is difficult to
derive accurate abundances when emission is included, since they depend
on the adopted central radiation source.
If, e.g., the blackbody temperature is taken equal to the dust sublimation
temperature of ![]() K, the inferred column densities increase
by only a factor of
K, the inferred column densities increase
by only a factor of ![]() 2.
2.
![\begin{figure}
\par\includegraphics[width=6.5cm,clip]{ms3426f6.eps}
\end{figure}](/articles/aa/full/2003/21/aa3426/Timg57.gif) |
Figure 6:
Gas/solid ratio for H2O versus
|
In Sect. 3 it is found that both the inferred gas-phase H2O abundances and the gas/solid ratios increase with the temperature of the warm gas, while the H2O ice abundances decrease. This indicates that H2O is a good tracer of the warm gas. Higher temperatures throughout the envelope are likely due to a higher ratio of the source luminosity to the envelope mass, which may correspond to dispersion of a larger fraction of the envelope (van der Tak et al. 2000b; van Dishoeck & van der Tak 2000). Thus, the fraction of warm over cold gas where H2O freezes-out is also increased. This suggests that the sources with higher H2O gas/solid ratios are more evolved than sources with low gas/solid ratios. The sources with the higher gas-phase H2O abundances and gas/solid ratios also show evidence for thermal processing of the ices (Smith et al. 1989; Boogert et al. 2000; Gerakines et al. 1999). Similarly, other evolutionary tracers, such as gas-phase HCN, C2H2, CH3OH, and CO2 show their highest abundances and/or gas/solid ratios for the same sources as gas-phase H2O and the lowest values for those sources where no gas-phase H2O has been detected (Lahuis & van Dishoeck 2000; van der Tak et al. 2000a; Boonman et al. 2003a). Together, this makes gas-phase H2O another useful tracer of the evolution of massive protostars.
Envelope models by Doty et al. (2002) and hot core models by
Charnley (1997) predict enhanced gas-phase H2O abundances
of up to ![]() 10-4 for
10-4 for ![]() -300 K in the case of pure gas-phase
chemistry. Similar abundances are predicted for
-300 K in the case of pure gas-phase
chemistry. Similar abundances are predicted for ![]() K if
ice evaporation is included. As noted in Sect. 3,
the increasing gas/solid ratios with temperature suggest that ice
evaporation is important for our sources.
The derived gas-phase H2O abundances in Table 2
are in agreement with these chemical models.
K if
ice evaporation is included. As noted in Sect. 3,
the increasing gas/solid ratios with temperature suggest that ice
evaporation is important for our sources.
The derived gas-phase H2O abundances in Table 2
are in agreement with these chemical models.
Shock models by Bergin et al. (1998) and Charnley & Kaufman
(2000) show that the H2O abundance in
post-shock gas can also reach values up
to ![]() 10-4. The low spectral resolution of the
ISO-SWS data does not allow to derive kinematic information from the
observed H2O lines, making it difficult to distinguish between
the envelope/hot core models and shock chemistry.
Similarly, a study of sulphur-bearing species, which are thought to
be good shock tracers, toward 8 of our sources cannot discriminate
between an origin in the shock or warm quiescent gas
(Keane et al. 2001a; van der Tak et al. 2003).
On the other hand, an analysis of gas-phase CO2 toward the same sources by
Boonman et al. (2003a) suggests that CO2 was destroyed
in
10-4. The low spectral resolution of the
ISO-SWS data does not allow to derive kinematic information from the
observed H2O lines, making it difficult to distinguish between
the envelope/hot core models and shock chemistry.
Similarly, a study of sulphur-bearing species, which are thought to
be good shock tracers, toward 8 of our sources cannot discriminate
between an origin in the shock or warm quiescent gas
(Keane et al. 2001a; van der Tak et al. 2003).
On the other hand, an analysis of gas-phase CO2 toward the same sources by
Boonman et al. (2003a) suggests that CO2 was destroyed
in ![]() -300 K gas by
the passage of a shock in the past and is either partially destroyed or
rapidly reformed through X-ray ionization in T>300 K gas.
The results from Sect. 4 do not provide strong
evidence for the destruction of H2O gas after evaporation, but
destruction of a small fraction of the gas-phase H2O in the warm interior
cannot be excluded at present. Thus, the inferred results for the
-300 K gas by
the passage of a shock in the past and is either partially destroyed or
rapidly reformed through X-ray ionization in T>300 K gas.
The results from Sect. 4 do not provide strong
evidence for the destruction of H2O gas after evaporation, but
destruction of a small fraction of the gas-phase H2O in the warm interior
cannot be excluded at present. Thus, the inferred results for the
![]() ro-vibrational band of gas-phase H2O are not inconsistent with the
gas-phase CO2 results from Boonman et al. (2003a).
High spectral-resolution observations, e.g. with the Heterodyne Instrument
for the Far-Infrared (HIFI) on board the Herschel
Space Observatory are needed to further investigate the origin of the abundant
gas-phase H2O in massive protostars.
ro-vibrational band of gas-phase H2O are not inconsistent with the
gas-phase CO2 results from Boonman et al. (2003a).
High spectral-resolution observations, e.g. with the Heterodyne Instrument
for the Far-Infrared (HIFI) on board the Herschel
Space Observatory are needed to further investigate the origin of the abundant
gas-phase H2O in massive protostars.
In addition to the high inferred gas-phase H2O abundances
from the infrared absorption bands, SWAS observations of
sources in our sample
indicate much lower H2O abundances of ![]() 10-9 to a
few
10-9 to a
few ![]() 10-8
in the outer region (Snell et al. 2000).
This suggests that a jump in the H2O abundance is present for our sources
and that the
ISO-SWS observations trace primarily the warmer gas in the inner envelope.
A similar jump is seen for CH3OH toward our sources
(van der Tak et al. 2000a).
In Paper II, the ISO-SWS observations presented here will be
combined with the pure rotational H2O lines observed with ISO-LWS and SWAS in order to find a chemical scenario that can explain all observed
H2O lines between 5 and 540
10-8
in the outer region (Snell et al. 2000).
This suggests that a jump in the H2O abundance is present for our sources
and that the
ISO-SWS observations trace primarily the warmer gas in the inner envelope.
A similar jump is seen for CH3OH toward our sources
(van der Tak et al. 2000a).
In Paper II, the ISO-SWS observations presented here will be
combined with the pure rotational H2O lines observed with ISO-LWS and SWAS in order to find a chemical scenario that can explain all observed
H2O lines between 5 and 540 ![]() m. In Paper II, the three above mentioned
processes that can produce high H2O abundances of 10-4,
i.e. high-T gas-phase chemistry, ice evaporation, and shocks, are
investigated in more detail.
m. In Paper II, the three above mentioned
processes that can produce high H2O abundances of 10-4,
i.e. high-T gas-phase chemistry, ice evaporation, and shocks, are
investigated in more detail.
ISO-SWS observations of the intermediate-mass protostars AFGL 490 and
AFGL 7009S show H2O abundances of a few ![]() 10-5
(Schreyer et al. 2002; Dartois et al. 1998),
while abundances of up to
10-5
(Schreyer et al. 2002; Dartois et al. 1998),
while abundances of up to ![]() 10-4 are found toward Orion-IRc2
(González-Alfonso et al. 1998; Harwit et al. 1998; Wright et al. 2000).
These are similar to the abundances inferred for the massive protostars
in Sect. 3.
Toward the low- to intermediate-mass star-forming regions
NGC 1333-IRAS 4 and IRAS 16293-2422
abundances of (3-
10-4 are found toward Orion-IRc2
(González-Alfonso et al. 1998; Harwit et al. 1998; Wright et al. 2000).
These are similar to the abundances inferred for the massive protostars
in Sect. 3.
Toward the low- to intermediate-mass star-forming regions
NGC 1333-IRAS 4 and IRAS 16293-2422
abundances of (3-
![]() for the outer and
(3-
for the outer and
(3-
![]() for the inner envelope have been derived indicating
a jump in the H2O abundance, similar to what is found for our sources
in Sect. 5.2 and Paper II
(Maret et al. 2002; Ceccarelli et al. 2000).
for the inner envelope have been derived indicating
a jump in the H2O abundance, similar to what is found for our sources
in Sect. 5.2 and Paper II
(Maret et al. 2002; Ceccarelli et al. 2000).
Acknowledgements
This work was supported by the NWO grant 614-41-003 and a NWO Spinoza grant. The authors would like to thank X. Tielens, R. Stark, and S. Doty for useful discussions.